via Northwestern University Feinberg School of Medicine
New technology may be applicable to other autoimmune diseases and allergies
Results of a new phase 2 clinical trial using technology developed at Northwestern Medicine show it is possible to induce immune tolerance to gluten in individuals with celiac disease. The findings may pave the way for treated celiac patients to eventually tolerate gluten in their diet.
After treatment with the technology, the patients were able to eat gluten with a substantial reduction in inflammation. The results also show a trend toward protecting patients’ small intestine from gluten exposure.
The findings will be presented as a late-breaking presentation Oct. 22 at the European Gastroenterology Week conference in Barcelona, Spain.
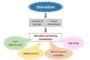
The technology is a biodegradable nanoparticle containing gluten that teaches the immune system the antigen (allergen) is safe. Thenanoparticle acts like a Trojan horse, hiding the allergen in a friendly shell, to convince the immune system not to attack it.
Beyond celiac disease, the finding sets the stage for the technology — a nanoparticle containing the antigen triggering the allergy or autoimmune disease — to treat a host of other diseases and allergies including multiple sclerosis, type 1 diabetes, peanut allergy, asthma and more.
The technology was developed in the lab of Stephen Miller, professor of microbiology and immunology at Northwestern University Feinberg School of Medicine, who has spent decades refining the technology.
“This is the first demonstration the technology works in patients,” said Miller, the Judy Gugenheim Research Professor of Microbiology and Immunology.“We have also shown that we can encapsulate myelin into the nanoparticle to induce tolerance to that substance in multiple sclerosis models, or put a protein from pancreatic beta cells to induce tolerance to insulin in type 1 diabetes models.”
When the allergen-loaded nanoparticle is injected into the bloodstream, the immune system isn’t concerned with it, because it sees the particle as innocuous debris. Then the nanoparticle and its hidden cargo are consumed by a macrophage, essentially a vacuum-cleaner cell that clears cellular debris and pathogens from the body.
“The vacuum-cleaner cell presents the allergen or antigen to the immune system in a way that says, ‘No worries, this belongs here,’” Miller said. “The immune system then shuts down its attack on the allergen, and the immune system is reset to normal.”
In the celiac disease trial, the nanoparticle was loaded with gliadin, the major component of dietary gluten, found in cereal grains such as wheat. A week after treatment, the patients were fed gluten for 14 days. Without treatment, celiac patients eating gluten developed marked immune responses to gliadin and damage in their small intestine. Celiac patients treated with the COUR nanoparticle, CNP-101, showed 90% less immune inflammation response than untreated patients. By stopping the inflammatory response, CNP-101 showed the capacity to protect the intestines from gluten related injury.
There currently is no treatment for celiac disease.
“Doctors can only prescribe gluten avoidance, which is not always effective and carries a heavy social and economic toll for celiac patients,” Miller said.
About 1% of the population has celiac disease,a serious autoimmune disease in which the ingestion of gluten leads to damage in the small intestine. When people with celiac disease eat gluten (a protein found in wheat), their body mounts an immune response that attacks the small intestine.
Autoimmune diseases generally can only be treated with immune suppressants that provide some relief, but undermine the immune system and lead to toxic side-effects. CNP-101 does not suppress the immune system but reverses the course of disease.
“Celiac disease is unlike many other autoimmune disorders because the offending antigen (environmental trigger) is well known — gluten in the diet,” said Dr. Ciaran Kelly, professor of medicine at Harvard Medical School and director of the Celiac Center at Beth Israel Deaconess Medical Center. “This makes celiac disease a perfect condition to address using this exciting nanoparticle induced immune tolerance approach.”
Kelly, who will be presenting the research abstract in Barcelona, has been working with Miller to apply the technology and define the therapeutic approach to treating celiac disease.
The nanotechnology was licensed to COUR Pharmaceuticals Co., a biotech based in Northbrook and co-founded by Miller. COUR developed CNP-101, which was granted Fast Track status from the U.S. Food and Drug Administration, and brought the therapy to patients in collaboration with Takeda Pharmaceuticals. Takeda will announce Tuesday they have acquired an exclusive global license to develop and commercialize this investigational medicine for celiac disease.
“Given the license by Takeda, COUR will focus on clinical programs in peanut allergy and multiple sclerosis in the near term and broaden even further over time,” said John J. Puisis, president and chief executive officer of COUR.
Learn more: New treatment may reverse celiac disease
The Latest on: Celiac disease
[google_news title=”” keyword=”celiac disease” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Celiac disease
- Best dry dog food for your pup, according to veterinarianson April 26, 2024 at 8:24 am
Did you adopt a dog or want to change your dog’s dry food brand? These veterinarians have advice on the best dry dog food to buy and which kibble to avoid.
- Microscopic Colitison April 25, 2024 at 2:29 pm
Microscopic colitis is inflammation of the colon's lining that causes chronic watery diarrhea. Thankfully, there are effective treatments available.
- Three Gluten-Free Friendly Restaurantson April 25, 2024 at 1:00 pm
And by far, the query that has appeared in my inbox with the greatest frequency has been about gluten-free dining. More than 2 million Americans have been diagnosed with Celiac — though researchers ...
- Sales of Gluten-Free Oats Global Market Strategic Growth & Forecast Analysis – 2023-2033on April 25, 2024 at 2:50 am
Gluten-Free Oats Market According to Future Market Insights (FMI), the gluten-free oats market valuation is likely to increase from US$ 168.3 million in 2023 to US$ 359.0 million by 2033. Overall ...
- First Wave BioPharma Announces Invited Paper for Special Edition on Celiac Disease in Peer-Reviewed Scientific Journal, Nutrientson April 24, 2024 at 6:00 am
The featured paper is entitled “Dynamics of Serologic Change to Gluten in Celiac Disease Patients” (doi.org/10.3390/nu15245083). The manuscript benefits from one of the largest databases of CeD ...
- Why Do I Keep Getting Canker Sores?on April 24, 2024 at 5:03 am
Certain foods, including spicy, acidic, or abrasive foods, can trigger pain in canker sores. Also, food allergies may trigger the development of a canker sore. Keeping a food diary can help you ...
- Why In-N-Out is the absolute best fast food chain for gluten-free dinerson April 24, 2024 at 5:01 am
Does In-N-Out Burger have gluten-free options? It does. Here's what to order if you're gluten-free, from animal-style burgers to fries and milkshakes.
- ‘Here’s How I Knew I Had Celiac Disease’: One Patient’s Storyon April 19, 2024 at 11:32 pm
Celiac disease affects around 3 million Americans, but fewer than one in three have been diagnosed, according to the Celiac Disease Foundation. The foundation defines celiac disease as a "genetic ...
- Pediatric Celiac Disease Diagnosis in Italy Shows Varied Diagnostic Delayson April 19, 2024 at 5:31 am
Younger Italian kids or those with family celiac history get quicker diagnoses; others with vague symptoms face delays.
- Immune-Mediated GI Disease May Up Eating Disorder Riskon April 18, 2024 at 11:24 pm
Researchers found a high prevalence of eating disorders in patients with major types of immune-mediated GI diseases, especially in pediatric patients diagnosed with ulcerative colitis.
via Bing News