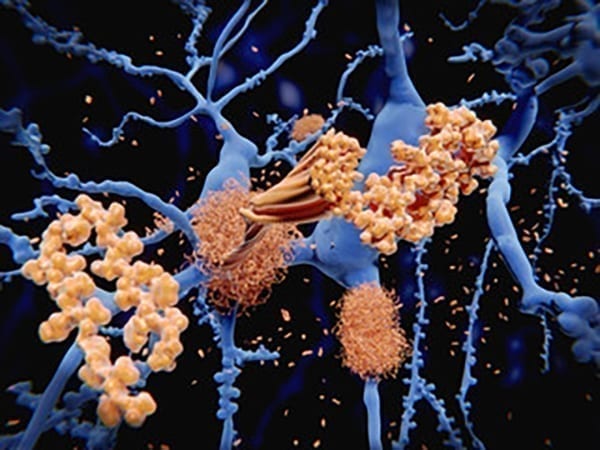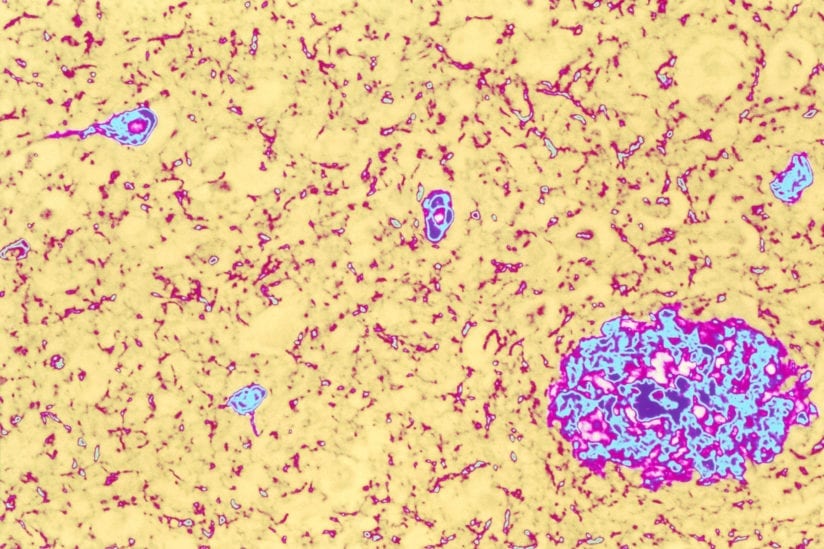
Colored light micrograph of brain tissue from an Alzheimer’s disease sufferer. Both of the classic pathological features of this disease are seen here. At lower right is a large plaque (pink/blue) containing the abnormal protein amyloid. Also seen are several neurofibrillary tangles (smaller blue areas), thickened parts of the cytoplasm of nerve cells. Both of these abnormalities disrupt the normal working of the brain. The symptoms of Alzheimer’s Disease include memory loss (particularly of recent events), disorientation and mood changes. Death occurs after several years of decline, and there is no cure. Magnification: x50 at 6x7cm size.
(Image/Simon Fraser, Science Source)
The power of simulations combined with experiments reveals why protein agglomerations typical of neurodegenerative diseases dissociate with infrared laser irradiation
The agglomeration of proteins into structures called amyloid plaques is a common feature of many neurodegenerative diseases, including Alzheimer’s. Now, scientists reveal, through experiments and simulations, how resonance with an infrared laser, when it is tuned to a specific frequency, causes amyloid fibrils to disintegrate from the inside out. Their findings open doors to novel therapeutic possibilities for amyloid plaque-related neurodegenerative diseases that have thus far been incurable.
A notable characteristic of several neurodegenerative diseases, such as Alzheimer’s and Parkinson’s, is the formation of harmful plaques that contain aggregates-also known as fibrils-of amyloid proteins. Unfortunately, even after decades of research, getting rid of these plaques has remained a herculean challenge. Thus, the treatment options available to patients with these disorders are limited and not very effective.
In recent years, instead of going down the chemical route using drugs, some scientists have turned to alternative approaches, such as ultrasound, to destroy amyloid fibrils and halt the progression of Alzheimer’s disease. Now, a research team led by Dr Takayasu Kawasaki (IR-FEL Research Center, Tokyo University of Science, Japan) and Dr Phuong H. Nguyen (Centre National de la Recherche Scientifique, France), including other researchers from the Aichi Synchrotron Radiation Center and the Synchrotron Radiation Research Center, Nagoya University, Japan, has used novel methods to show how infrared-laser irradiation can destroy amyloid fibrils.
In their study,published in Journal of Physical Chemistry B, the scientists present the results of laser experiments and molecular dynamics simulations. This two-pronged attack on the problem was necessary because of the inherent limitations of each approach, as Dr Kawasaki explains, “While laser experiments coupled with various microscopy methods can provide information about the morphology and structural evolution of amyloid fibrils after laser irradiation, these experiments have limited spatial and temporal resolutions, thus preventing a full understanding of the underlying molecular mechanisms. On the other hand, though this information can be obtained from molecular simulations, the laser intensity and irradiation time used in simulations are very different from those used in actual experiments. It is therefore important to determine whether the process of laser-induced fibril dissociation obtained through experiments and simulations is similar.”
The scientists used a portion of a yeast protein that is known to form amyloid fibrils on its own. In their laser experiments, they tuned the frequency of an infrared laser beam to that of the “amide I band” of the fibril, creating resonance. Scanning electron microscopy images confirmed that the amyloid fibrils disassembled upon laser irradiation at the resonance frequency, and a combination of spectroscopy techniques revealed details about the final structure after fibril dissociation.
For the simulations, the researchers employed a technique that a few members of the current team had previously developed, called “nonequilibrium molecular dynamics (NEMD) simulations.” Its results corroborated those of the experiment and additionally clarified the entire amyloid dissociation process down to very specific details. Through the simulations, the scientists observed that the process begins at the core of the fibril where the resonance breaks intermolecular hydrogen bonds and thus separates the proteins in the aggregate. The disruption to this structure then spreads outward to the extremities of the fibril.
Together, the experiment and simulation make a good case for a novel treatment possibility for neurodegenerative disorders. Dr Kawasaki remarks, “In view of the inability of existing drugs to slow or reverse the cognitive impairment in Alzheimer’s disease, developing non-pharmaceutical approaches is very desirable. The ability to use infrared lasers to dissociate amyloid fibrils opens up a promising approach.”
The team’s long-term goal is to establish a framework combining laser experiments with NEMD simulations to study the process of fibril dissociation in even more detail, and new works are already underway. All these efforts will hopefully light a beacon of hope for those dealing with Alzheimer’s or other neurodegenerative diseases.
The Latest Updates from Bing News & Google News
Go deeper with Bing News on:
Amyloid plaques
- Unlocking Alzheimer’s: New Study Reveals Early Stage Mechanisms
APP-CTFs interfere with the communication between organelles, disrupting cellular balance. Alzheimer's disease (AD) continues to be a significant and widespread neurodegenerative condition, impacting ...
- UF Health begins offering new Alzheimer’s drug
University of Florida Health is offering a new drug therapy that could temporarily slow the progression of Alzheimer’s disease in some patients.
- Georgia Tech researchers study pulsing light as non-invasive Alzheimer's disease treatment
Researchers at Georgia Tech are studying a novel way to help people living with Alzheimer's disease: using pulsing light and buzzing sounds.
- New Alzheimer’s Treatment Uses Light & X-Rays To Hit Harmful Protein
A new treatment for early-stage Alzheimer’s uses nanoparticles and light to slow the progress of the disease, set off by a low-powered x-ray.
- The #1 Food to Limit to Reduce Your Risk of Dementia, According to Dietitians
Taking steps to reduce dementia risk is one positive step for brain health. While there isn’t one food that will cause dementia, sugary candy tops the list of foods that are recommended to be limited ...
Go deeper with Google Headlines on:
Amyloid plaques
[google_news title=”” keyword=”amyloid plaques” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]
Go deeper with Bing News on:
Laser irradiation
- Florida teen arrested for allegedly pointing laser at sheriff's helicopter: 'He's blinding our pilot'
A 13-year-old Florida boy is charged with a felony after allegedly pointing a laser at a sheriff's department helicopter. The Pinellas County Sheriff’s Office said the helicopter was in the sky ...
- The Best At-Home Laser Hair Removal Device for Professional Looking Results
This content is created independently from TIME’s editorial staff. Learn more about it. At-home laser hair removal devices are suitable alternatives to in-office treatments for people looking ...
- 4 of the Best Home Laser Hair Removal Devices by Hair Color, Coarseness, and More
Here’s our process. You used to have to visit a dermatologist for laser hair removal, and this is still the most effective route, but you can consider home laser hair removal devices instead.
- Best laser printers of 2024
Looking for the best laser printers? Our team of reviewers tested out top models from Xerox, Brother, Lexmark, and more to find out which are worth taking up office space. If you print a lot of ...
- Best laser engravers of 2024
Looking for the best laser engravers? We went hands-on with some of the top models to find out which ones are a cut above the rest. Like the best 3D printers we tested, laser engraving machines ...
Go deeper with Google Headlines on:
Laser irradiation
[google_news title=”” keyword=”laser irradiation” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]