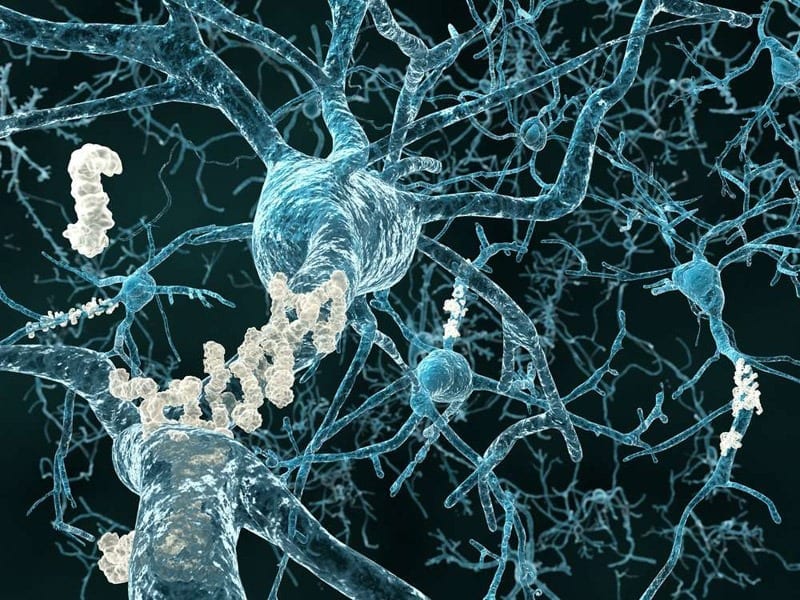
Aducanumab, an antibody developed by the University of Zurich, has been shown to trigger a meaningful reduction of harmful beta-amyloid plaques in patients with early-stage Alzheimer’s disease.
These protein deposits in the brain are a classic sign of Alzheimer’s disease and contribute to the progressive degeneration of brain cells. The researchers furthermore demonstrated in an early stage clinical study that, after one year of treatment with Aducanumab, cognitive decline could be significantly slowed in antibody-treated patients as opposed to the placebo group.
Although the causes of Alzheimer’s disease are still unknown, it is clear that the disease commences with progressive amyloid deposition in the brains of affected persons between ten and fifteen years before the emergence of initial clinical symptoms such as memory loss. Researchers have now been able to show that Aducanumab, a human monoclonal antibody, selectively binds brain amyloid plaques, thus enabling microglial cells to remove the plaques. A one-year treatment with the antibody, as part of a phase Ib study, resulted in almost complete clearance of the brain amyloid plaques in the study group patients. The results, which were realized by researchers at UZH together with the biotech company “Biogen” and the UZH spin-off “Neurimmune,” have been published in the renowned science journal “Nature.”
Reduction of brain amyloid plaque is dependent on treatment duration and dosage
“The results of this clinical study make us optimistic that we can potentially make a great step forward in treating Alzheimer’s disease,” says Roger M. Nitsch, professor at the Institute for Regenerative Medicine at UZH. “The effect of the antibody is very impressive. And the outcome is dependent on the dosage and length of treatment.” After one year of treatment, practically no beta-amyloid plaques could be detected in the patients who received the highest dose of the antibody.
The antibody was developed with the help of a technology platform from “Neurimmune.” Using blood collected from elderly persons aged up to one hundred and demonstrating no cognitive impairment, the researchers isolated precisely those immune cells whose antibodies are able to identify toxic beta-amyloid plaques but not the amyloid precursor protein that is present throughout the human body and that presumably plays an important role in the growth of nerve cells. The good safety profile of Aducanumab in patients may well be attributed to the antibody’s specific capacity to bond with the abnormally folded beta-amyloid protein fragment as well as the fact that the antibody is of human origin.
Investigational treatment also curbs cognitive decline
165 patients with early-stage Alzheimer’s disease were treated in the phase 1b clinical trial. Although not initially planned as a primary study objective, the good results encouraged researchers to additionally investigate how the treatment affected the symptoms of disease. This was evaluated via standardized questionnaires to assess the cognitive abilities and everyday activities of the patients. “Aducanumab also showed positive effects on clinical symptoms,” is how Nitsch sums up the findings. “While patients in the placebo group exhibited significant cognitive decline, cognitive ability remained distinctly more stable in patients receiving the antibody.”
Some of the trial participants temporarily suffered from amyloid-related imaging abnormality (ARIA), an adverse effect that can be detected via magnetic resonance imaging. In a minority of cases, this was accompanied by temporary mild to moderate headaches. The UZH researchers believe that ARIA is a measurable biological effect of amyloid clearance.
The promising effects of Aducanumab are currently being investigated in two large phase three clinical studies to further evaluate safety and efficacy. Involving over 300 centers in 20 countries throughout North America, Europe, and Asia, these studies are evaluating the effectiveness and safety of the antibody on a total of 2,700 patients with early-stage Alzheimer’s disease.
Learn more: Antibody Reduces Harmful Brain Amyloid Plaques in Alzheimer’s Patients
The Latest on: Aducanumab
[google_news title=”” keyword=”Aducanumab” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Aducanumab
- Jay Leno and wife Mavis attend ‘Unfrosted’ red carpet event amidst her dementia diagnosison May 1, 2024 at 8:01 am
TREATMENTS AND SYMPTOMS TO WATCH OUT FOR. IT’S A NEW AGE IN ALZHEIMER’S TREATMENT. TWO NEW DRUGS THAT SLOW THE DISEASE ADUCANUMAB BRAND NAME ADUHELM APPROVED BY THE FDA IN 2021 AND LECANEMAB BRAND ...
- Alzheimer's drug development pipeline: Positive results, new insight on biomarkers position 2024 as 'learning year'on April 24, 2024 at 12:13 pm
The world of Alzheimer's treatments is at an inflection point as more potential drugs make their way out of clinical trials.
- People’s Pharmacy: Can toothpaste contribute to mouth sores?on April 21, 2024 at 9:04 pm
A dental hygienist said that canker sores are caused by sensitivity to sodium lauryl sulfate, a foaming agent present in many but not all toothpastes.
- What to know about medications for memory losson April 3, 2024 at 5:00 pm
Aducanumab is the first-line treatment for people with early stage Alzheimer’s disease and is available in the United States under the brand name Aduhelm. Doctors may also prescribe it for other ...
- One step back: Why the new Alzheimer's plaque-attack drugs don't workon March 14, 2024 at 10:04 am
Aducanumab has netted a mere $3 million in sales to date, implying that fewer than 100 people have reached into their pockets to pay the full price since its approval. Whatever.
- I took the now-defunct Alzheimer's medication for seven years and it 'paused' my disease - without it there's no hopeon February 25, 2024 at 5:06 am
In 2012, Geri Taylor was a top executive at a large long-term care facility in New York City when she began to experience signs of cognitive decline. On one occasion, the 69 year-old lost her ...
- Pharmaceutical Industry Ought To Justify Drug Prices On Value, Not R&D Costson June 1, 2023 at 4:10 am
The Aduhelm (aducanumab) debacle, for example, shows that products with marginal value, or perhaps even no incremental value, don’t fetch a price (or reimbursement) they `deserve’ based on all ...
- Aducanumab: Scots could be among first to trial Alzheimer’s drugon November 18, 2021 at 9:00 pm
The European Medicines Agency (EMA) looks set to reject the drug Aducanumab next month for widespread use in countries including the UK. However a leading Scots dementia expert says that decision ...
- Biogen's Alzheimer's Drug Gets Negative European Voteon November 17, 2021 at 5:45 am
Biogen (BIIB) shares fell Wednesday after the pharmaceutical titan reported a negative regulatory vote in Europe on its Alzheimer’s treatment, aducanumab. The Committee for Medicinal ...
via Bing News