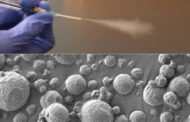
MIT engineers have designed coiled “nanoyarn,” shown as an artist’s interpretation here. The twisted fibers are lined with living cells and may be used to repair injured muscles and tendons while maintaining their flexibility.
Image: Felice Frankel
Twisted fibers coated with living cells could assist healing of injured muscles and tendons.
The human body is held together by an intricate cable system of tendons and muscles, engineered by nature to be tough and highly stretchable. An injury to any of these tissues, particularly in a major joint like the shoulder or knee, can require surgical repairs and weeks of limited mobility to fully heal.
Now MIT engineers have come up with a tissue engineering design that may enable flexible range of motion in injured tendons and muscles during healing.
The team has engineered small coils lined with living cells, that they say could act as stretchy scaffolds for repairing damaged muscles and tendons. The coils are made from hundreds of thousands of biocompatible nanofibers, tightly twisted into coils resembling miniature nautical rope, or yarn.
The researchers coated the yarn with living cells, including muscle and mesenchymal stem cells, which naturally grow and align along the yarn, into patterns similar to muscle tissue. The researchers found the yarn’s coiled configuration helps to keep cells alive and growing, even as the team stretched and bent the yarn multiple times.
In the future, the researchers envision doctors could line patients’ damaged tendons and muscles with this new flexible material, which would be coated with the same cells that make up the injured tissue. The “yarn’s” stretchiness could help maintain a patient’s range of motion while new cells continue to grow to replace the injured tissue.
“When you repair muscle or tendon, you really have to fix their movement for a period of time, by wearing a boot, for example,” says Ming Guo, assistant professor of mechanical engineering at MIT. “With this nanofiber yarn, the hope is, you won’t have to wearing anything like that.”
Guo and his colleagues published their results this week in the Proceedings of the National Academy of Sciences. His MIT co-authors are Yiwei Li, Yukun Hao, Satish Gupta, and Jiliang Hu. The team also includes Fengyun Guo, Yaqiong Wang, Nü Wang, and Yong Zhao, of Beihang University.
Stuck on gum
The new nanofiber yarn was inspired in part by the group’s previous work on lobster membranes, where they found the crustacean’s tough yet stretchy underbelly is due to a layered, plywood-like structure. Each microscopic layer contains hundreds of thousands of nanofibers, all aligned in the same direction, at an angle that is slightly offset from the layer just above and below.
The nanofibers’ precise alignment makes each individual layer highly stretchable in the direction in which the fibers are arranged. Guo, whose work focuses on biomechanics, saw the lobster’s natural stretchy patterning as an inspiration for designing artificial tissues, particularly for high-stretch regions of the body such as the shoulder and knee.
Guo says biomedical engineers have embedded muscle cells in other stretchy materials such as hydrogels, in attempts to fashion flexible artificial tissues. However, while the hydrogels themselves are stretchy and tough, the embedded cells tend to snap when stretched, like tissue paper stuck on a piece of gum.
“When you largely deform a material like hydrogel, it will be stretched just fine, but the cells can’t take it,” Guo says. “A living cell is sensitive, and when you stretch them, they die.”
Shelter in a slinky
The researchers realized that simply considering the stretchability of a material would not be enough to design an artificial tissue. That material would also have to be able to protect cells from the severe strains produced when the material is stretched.
The team looked to actual muscles and tendons for further inspiration, and observed that the tissues are made from strands of aligned protein fibers, coiled together to form microscopic helices, along which muscle cells grow. It turns out that, when the protein coils stretch out, the muscle cells simply rotate, like tiny pieces of tissue paper stuck on a slinky.
Guo looked to replicate this natural, stretchy, cell-protecting structure as an artificial tissue material. To do so, the team first created hundreds of thousands of aligned nanofibers, using electrospinning, a technique that uses electric force to spin ultrathin fibers out from a solution of polymer or other materials. In this case, he generated nanofibers made from biocompatible materials such as cellulose.
The team then bundled aligned fibers together and twisted them slowly to form first a spiral, and then an even tighter coil, ultimately resembling yarn and measuring about half a millimeter wide. Finally, they seeded live cells along each coil, including muscle cells, mesenchymal stem cells, and human breast cancer cells.
The researchers then repeatedly stretched each coil up to six times its original length, and found that the majority of cells on each coil remained alive and continued to grow as the coils were stretched. Interestingly, when they seeded cells on looser, spiral-shaped structures made from the same materials, they found cells were less likely to remain alive. Guo says the structure of the tighter coils seems to “shelter” cells from damage.
Going forward, the group plans to fabricate similar coils from other biocompatible materials such as silk, which could ultimately be injected into an injured tissue. The coils could provide a temporary, flexible scaffold for new cells to grow. Once the cells successfully repair an injury, the scaffold can dissolve away.
“We may be able to one day embed these structures under the skin, and the [coil] material would eventually be digested, while the new cells stay put,” Guo says. “The nice thing about this method is, it’s really general, and we can try different materials. This may push the limit of tissue engineering a lot.”
Learn more: “Nanofiber yarn” makes for stretchy, protective artificial tissue
The Latest on: Tissue engineering
[google_news title=”” keyword=”tissue engineering” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Tissue engineering
- National Science Foundation Awards $450-K Grant to ASU’s Engineering Programon April 25, 2024 at 10:15 am
The National Science Foundation (NSF) has just awarded Alabama State University's Dr. Vineeth Vijayan -- an assistant professor in the University's engineering department -- a grant of $450,000. He ...
- New experimental evidence unlocks a puzzle in vascular tissue engineeringon April 25, 2024 at 9:25 am
Angiogenesis is a process of forming hierarchical vascular networks in living tissues. Its complexity makes the controlled generation of blood vessels in laboratory conditions a highly challenging ...
- Lab-made miniorgans take scientists a step closer to curing canceron April 25, 2024 at 8:34 am
In the fight to cure cancer, scientists need better models. And they might just get them — human-derived organoids — thanks to light.
- Mini Colons Mimic Healthy, Tumor Tissue In Colorectal Cancer Developmenton April 25, 2024 at 12:01 am
The mini colons replicate both the physical structure and cellular diversity in the colon in both healthy and diseased states.
- Tissue Engineering and Regeneration Market [2028]: Navigating Opportunities and Challengeson April 24, 2024 at 3:11 am
USD 14.57 billion in 2022 and is poised for substantial growth, projecting a Compound Annual Growth Rate (CAGR) of 8.35% through 2028.
- Scientists develop maleic acid-treated bacterial cellulose gel for enhancing bone repairon April 15, 2024 at 1:48 pm
The field of bone tissue engineering (BTE) is a promising avenue for addressing bone injuries and defects by constructing artificial scaffolds with bionic functionalities. Due to its unique 3D network ...
- Team replicates an adult human ear using tissue engineering, 3D printingon April 15, 2024 at 3:33 am
The sterilized cartilage was placed on ear-shaped plastic scaffolds created on a 3D printer, featuring the dimensions and curves of the person’s ear. The researchers explained that the small pieces of ...
- Advancing tissue engineering with shape memory hydrogelson March 13, 2024 at 7:55 am
On seeding the hydrogel with mouse embryo fibroblast (NIH/3T3) cells, the researchers observed a direct correlation between the elastic modulus of the hydrogel and the number of adhered cells. In ...
- Cardiovascular Reparative Medicine and Tissue Engineeringon February 20, 2024 at 2:49 pm
Cardiovascular Reparative Medicine and Tissue Engineering (CRMTE) aims to develop future technologies and therapeutic strategies that will serve as treatment for cardiovascular disease. CRMTE includes ...
- TissueFab® Sacrificial bioink for tissue engineering applicationson November 14, 2023 at 4:00 am
Polaxomer is a thermos-responsive hydrogel that has been employed in bioprinting and tissue engineering as a mold, track patterning, and sacrificial material. Due to the nature of micellar-packing ...
via Bing News