via Phys.org
An international research team led by the Institute of Bioengineering and Nanotechnology (IBN) of the Agency for Science, Technology and Research (A*STAR) and IBM Research developed a synthetic molecule that can kill five deadly types of multidrug-resistant bacteria with limited, if any, side effects. Their new material could be developed into an antimicrobial drug to treat patients with antibioticresistant infections.
This finding was reported in the scientific journal Nature Communications.
Superbugs that are resistant to antibiotics are a serious health threat. According to the UK Review on Antimicrobial Resistance, superbugs kill around 700,000 people worldwide each year. By 2050, 10 million people could die each year if existing antibiotics continue to lose their effectiveness.
“There is an urgent global need for new antimicrobials that are effective against superbugs. The situation has become more acute because bacteria are starting to develop resistance to the last-line antibiotics, which are given only to patients infected with bacteria resistant to available antibiotics,” said Professor Jackie Y. Ying, Executive Director of IBN.
The research community is trying to develop alternatives to antibiotics using synthetic polymers. However, the antimicrobial polymers developed so far are either too toxic for clinical use, not biodegradable or can only target one type of bacteria.
To address this problem, Dr Yi Yan Yang from IBN brought together a multidisciplinary research team from the US, China and Singapore to develop a new class of antimicrobial polymers called guanidinium-functionalized polycarbonates with a unique killing mechanism that can target a broad range of multidrug-resistant bacteria. It is biodegradable and non-toxic to human cells.
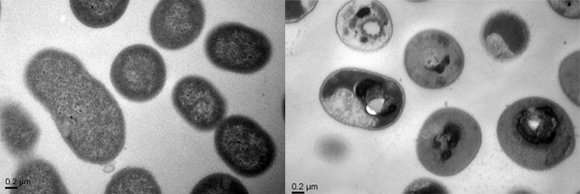
The polymer kills bacteria in the following way. First, the polymer binds specifically to the bacterial cell. Then, the polymer is transported across the bacterial cell membrane into the cytoplasm, where it causes precipitation of the cell contents (proteins and genes), resulting in cell death.
The team tested the polymers on mice infected with five hard-to-treat multidrug-resistant bacteria: Acinetobacter baumannii, Escherichia coli, Klebsiella pneumoniae, methicillinresistant Staphylococcus aureu and Pseudomonas aeruginosa. These superbugs are commonly acquired by patients in the hospitals and can cause systemic infections that lead to septic shock and multiple organ failure. The results showed that the bacteria were effectively removed from the mice and no toxicity was observed.
The researchers then further tested the effectiveness of the polymers on mice with two types of systemic infections caused by superbugs: peritonitis (an infection of the stomach’s inner lining) and lung infections from Pseudomonas aeruginosa. The polymers eliminated the bacterial infections in both groups of mice with negligible toxicity.
Dr Yi Yan Yang, Group Leader at IBN said, “We have demonstrated the first example of a biodegradable synthetic macromolecule with broad-spectrum antimicrobial activity in mice, unique killing mechanism and no toxicity. Once the polymer finishes its job of killing the bacteria, it will be naturally degraded after three days and will not remain in the body. This antimicrobial agent shows great promise for the treatment and prevention of multidrug-resistant systemic infections.”
“This study illustrates the potential for this new research field we denote as ‘macromolecular therapeutics’ to create entirely new classes of treatments for multiple diseases,” said Dr James Hedrick, Distinguished Research Staff Member, IBM Research – Almaden, San Jose, California. “In 2016, we demonstrated the efficacy of synthetic polymers to combat deadly viral diseases. The current research for treating bacterial infections rounds out our ability to someday treat a spectrum of infectious diseases with a single, new type of mechanism without the onset of resistance.”
To determine whether the bacteria will develop any resistance to the polymer, the team collaborated with Dr Paola Florez de Sessions at A*STAR’s Genome Institute of Singapore and the Cell Engineering group of Dr Simone Bianco at IBM Research – Almaden to perform genomic analysis. They found that the bacteria did not show any resistance development even after multiple treatments with the polymer.
Learn more: New Molecule Can Kill Five Types of Deadly Drug-Resistant Superbugs
The Latest on: Antimicrobial drug
[google_news title=”” keyword=”Antimicrobial drug” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Antimicrobial drug
- AI Model Shows Promise in Fighting Drug Resistanceon April 26, 2024 at 8:31 pm
As antibiotic resistance continues to pose a formidable challenge, AI-driven solutions offer a beacon of hope in the fight against drug-resistant pathogens.
- Breakthrough Vaccine Shows High Protection Against Drug-Resistant Superbug in Miceon April 26, 2024 at 4:03 pm
Scientists have developed a vaccine against a notorious drug-resistant superbug, targeting molecules on its surface that are also found on other bacteria and fungi.
- Vaccines have a crucial role in tackling antimicrobial resistanceon April 26, 2024 at 10:04 am
Antimicrobial resistance is a leading cause of death and disability worldwide. In 2019, it caused over 1 million deaths globally and was linked to almost 5 million.
- Common antibiotic Neosporin may shield against viral respiratory infectionson April 26, 2024 at 9:00 am
A study conducted first in mice and then in human volunteers suggests that a common antibiotic, neomycin, which is the main active ingredient in Neosporin, may help protect against viral respiratory ...
- Study: Antibiotic use in moderate COVID-19 linked to clinical deteriorationon April 26, 2024 at 7:56 am
Antibiotic treatment of adults hospitalized with moderate COVID-19 is associated with clinical deterioration, despite the drugs being given to over 40% of patients, according to new research being ...
- What to Know About Pivya, the New Antibiotic Treatment for UTIson April 26, 2024 at 4:06 am
The Food and Drug Administration recently approved Pivya (pivmecillinam) to treat uncomplicated UTIs in female adults. Clinical trials show that Pivya is effective and side effects are limited.
- Climate change fuels the rise of global antimicrobial resistanceon April 25, 2024 at 10:34 am
Climate change exacerbates antimicrobial resistance by altering environments where microbes thrive, leading to increased disease spread.
- FDA approves new antibiotic against UTIson April 25, 2024 at 8:10 am
"The FDA is committed to fostering new antibiotic availability when they prove to be safe and effective, and Pivya will provide an additional treatment option for uncomplicated UTIs," he added.
- What We’re Reading: FDA Approves UTI Antibiotic; Ozempic, Wegovy Price Investigation; US Births Fallon April 25, 2024 at 8:07 am
The FDA recently approved an antibiotic for the treatment of urinary tract infections (UTIs) in women; a Senate committee recently launched an investigation into the prices of Novo Nordisk’s diabetes ...
- F.D.A. Approves Antibiotic for Increasingly Hard-to-Treat Urinary Tract Infectionson April 24, 2024 at 1:45 pm
Pivmecillinam, which has been used in Europe for decades, will become available next year to women 18 and older.
via Bing News