An international research team is perfecting a method to predict the potential clinical implications of new drugs before clinical trials even start.
An international research team has developed a new strategy that can predict the potential clinical implications of new therapeutic compounds based on simple cellular responses. This discovery was partly led by scientists affiliated with Université de Montréal (UdeM), and represents a major step forward in developing more effective drugs with fewer side effects, much faster than before. The researchers conducted their work at Centre de Recherche de l’Hôpital Ste-Justine and published their findings in the prestigious journal Nature Communications.
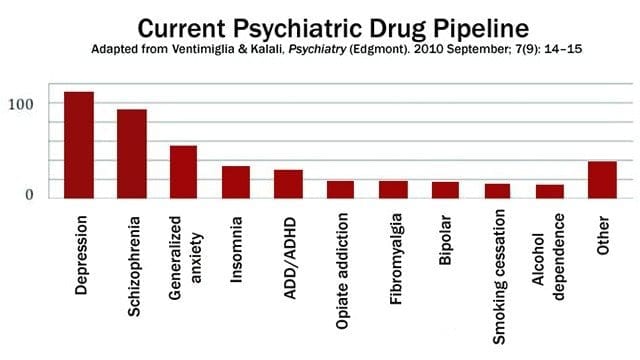
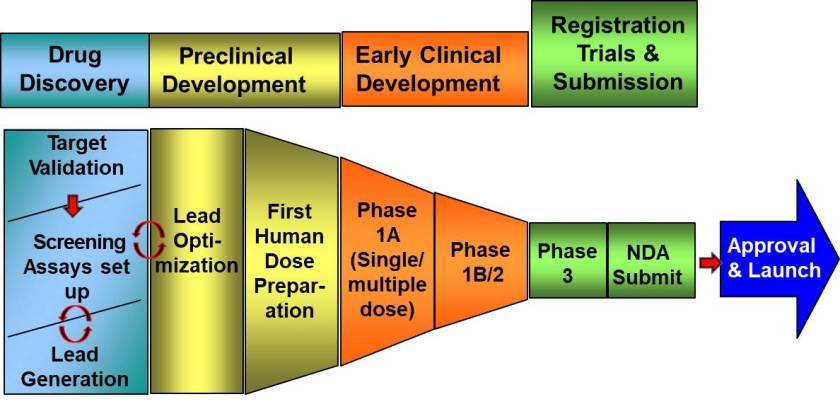
Developing new drugs is a long, complex and costly process. It starts with identifying the molecule or “ligand” (such as a drug, hormone or neurotransmitter) that can activate or block the target or “receptor” involved in a disease. Compound identification and validation is one of the most important steps in ensuring that a new drug provides an effective clinical response with the fewest possible side effects.
“Most new drugs tested on human subjects fail in clinical trials because the therapeutic response is insufficient. Developing a strategy that infers potential clinical responses early in the drug discovery process would significantly improve drug candidate selection,” said Besma Benredjem, the study’s co-lead author and a doctoral student in pharmacology at UdeM.
Finding the needle in a haystack
“Our main goal was finding a way to categorize a large number of drug candidates based on similarities in their effectiveness in triggering a multiplicity of cellular responses that help identify the therapeutic action of new compounds,” said Professor Graciela Piñeyro, co-senior author of the study and a researcher at CHU Sainte-Justine. To accomplish this, she worked with Dr. Olivier Lichtarge of Baylor College of Medicine, who uses advanced bioinformatic analysis to compare and group ligands according to fairly comprehensive signalling profiles.
Drugs produce desired or undesired clinical actions by changing basic signals within cells. By grouping drugs with known clinical actions and new ligands, we can infer the clinical actions of new compounds by comparing the similarities and differences in their signals with known drugs to promote desired clinical responses and avoid side effects.
This method of analysis was developed by using opioid analgesics as prototypes. This made it possible for the team to associate simple cellular signals produced by opioids such as oxycodone, morphine and fentanyl with the frequency with which respiratory depression and other undesirable side effects of these drugs were reported to the Food and Drug Administration’s pharmacovigilance program. At the height of the opioid epidemic, when the risk of death by respiratory depression is at its highest, the team believes this new analytical strategy could lead to the development of safer opioids.
“Thanks to our findings, we can now classify a large number of compounds while taking a multitude of cellular signals into account. The wealth of comparisons this provides increases this classification’s predictive value for clinical responses,” said Professor Michel Bouvier, the study’s co-senior author and a principal investigator of molecular pharmacology and Chief Executive Officer of UdeM’s Institute for Research in Immunology and Cancer. “We think we can help patients by speeding up the drug discovery process so clinical trials can start earlier.”
“Our next goal is to use a similar approach to test cannabis products that may produce harmful neuropsychiatric actions among young people, and identify which cannabis extracts are most effective at treating chronic pain,” added Besma Benredjem.
Learn more: Speeding up the drug discovery process to help patients
The Latest on: Drug development
[google_news title=”” keyword=”drug development” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Drug development
- Delphia starts up with $67M to make a new type of cancer drugon May 4, 2024 at 4:01 pm
The startup is developing medicines designed to overstress cellular pathways to the point tumor cells die, an approach the biotech calls “activation lethality.” ...
- Amgen soars 16% after it talks up a new weight loss drug that could be injected monthly instead of weeklyon May 3, 2024 at 9:46 am
Goldman Sachs said Amgen's experimental GLP-1 drug could have peak sales of $5 billion, or 5% of the estimated $100 billion weight loss drug market.
- How AI Could Transform Drug Development And The Life Scienceson May 2, 2024 at 11:45 am
The implementation of AI in healthcare spans from developing drugs to using AI in the real world. This panel from Imagination In Action's 'Forging the Future of Business with AI' Summit talks about ...
- Deeper understanding of malaria parasite sexual development unlocks opportunities to block disease spreadon May 2, 2024 at 11:00 am
For the first time, the developmental stages of the deadliest human malaria parasite have been mapped in high resolution, allowing researchers to understand this ever-adapting adversary in more detail ...
- Project Optimus: helping or hindering cancer drug development?on May 2, 2024 at 1:01 am
Experts say that Project Optimus is a necessary reform in the oncology space, but one that should be applied with caution.
- Global Functional Dyspepsia Drug Market to reach a valuation of US$ 16.4 billion by 2034, Future Market Insights, Inc. Projectionon May 1, 2024 at 7:48 am
General Anesthesia Drugs Market Size: The global general anesthesia drugs market size is estimated at US$ 4,978.1 million in 2023. Projections indicate a steady growth with a steady CAGR of 3.3% ...
- Harmony Biosciences shares soar on acquisition of New Jersey epilepsy drug developeron April 30, 2024 at 8:31 am
The deal consists of an upfront payment of $35 million and potential milestone payments of up to $645 million.
- A Pharmaceutical Company is Working to Speed Up the Drug Development Process with AIon April 28, 2024 at 9:50 pm
Algorae Pharmaceuticals is finding ways to revamp its process of drug development from cost saving and speed to timeline of drug research and delivery by ...
- Alzheimer's drug development pipeline: Positive results, new insight on biomarkers position 2024 as 'learning year'on April 24, 2024 at 12:13 pm
The world of Alzheimer's treatments is at an inflection point as more potential drugs make their way out of clinical trials.
- Collaboration and Partnerships in Accelerating Cancer Drug Developmenton April 23, 2024 at 5:53 am
Lorna Rothery spoke to the Cancer Drug Development Forum’s Managing Director, Professor Jaap Verweij and Chairperson of the CDDF Board of Directors, Professor Ruth Plummer, about opportunities and cha ...
via Bing News