Causes of hydrogen generation and incessant accumulation within the cell in the aqueous rechargeable batteries
CREDIT: Korea Institute of Science and Technology
Automatic conversion of hydrogen gas into water makes batteries safer. A breakthrough technology for the commercialization of cheaper, safer aqueous rechargeable batteries.
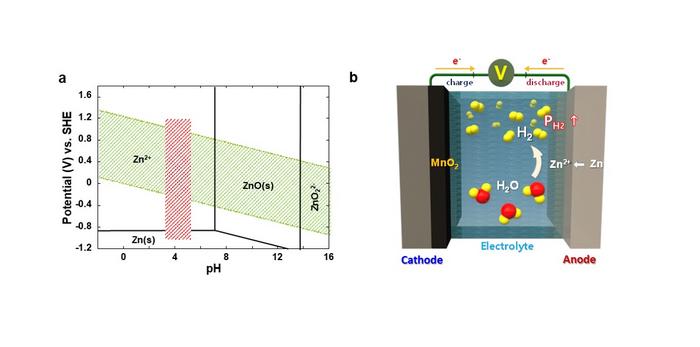
This summer, the planet is suffering from unprecedented heat waves and heavy rainfalls. Developing renewable energy and expanding associated infrastructure has become an essential survival strategy to ensure the sustainability of the planet in crisis, but it has obvious limitations due to the volatility of electricity production, which relies on uncertain variables like labile weather conditions. For this reason, the demand for energy storage systems (ESS) that can store and supply electricity as needed is ever-increasing, but lithium-ion batteries (LIBs) currently employed in ESS are not only highly expensive, but also prone to potential fire, so there is an urgent need to develop cheaper and safer alternatives.
A research team led by Dr. Oh, Si Hyoung of the Energy Storage Research Center at the Korea Institute of Science and Technology (KIST) has developed a highly safe aqueous rechargeable battery that can offer a timely substitute that meets the cost and safety needs. Despite of lower energy density achievable, aqueous rechargeable batteries have a significant economic advantage as the cost of raw materials is much lower than LIBs. However, inveterate hydrogen gas generated from parasitic water decomposition causes a gradual rise in internal pressure and eventual depletion of the electrolyte, which poses a sizeable threat on the battery safety, making commercialization difficult.
Until now, researchers have often tried to evade this issue by installing a surface protection layer that minimizes the contact area between the metal anode and the electrolyte. However, the corrosion of the metal anode and accompanying decomposition of water in the electrolyte is inevitable in most cases, and incessant accumulation of hydrogen gas can cause a potential detonation in long-term operation.
To cope with this critical issue, the research team has developed a composite catalyst consisting of manganese dioxide and palladium, which is capable of automatically converting hydrogen gas generated inside the cell into water, ensuring both the performance and safety of the cell. Manganese dioxide does not react with hydrogen gas under normal circumstances, but when a small amount of palladium is added, hydrogen is readily absorbed by the catalysts, being regenerated into water. In the prototype cell loaded with the newly developed catalysts, the internal pressure of the cell was maintained well below the safety limit, and no electrolyte depletion was observed.
The results of this research effectively solves one of the most concerning safety issues in the aqueous batteries, making a major stride towards commercial application to ESS in the future. Replacing LIBs by cheaper and safer aqueous batteries can even trigger a rapid growth of global market for ESS.
“This technology pertains to a customized safety strategy for aqueous rechargeable batteries, based on the built-in active safety mechanism, through which risk factors are automatically controlled.” said Dr. Oh, Si Hyoung of KIST. “Moreover, it can be applied to various industrial facilities where hydrogen gas leakage is one of major safety concerns (for instance, hydrogen gas station, nuclear power plant etc) to protect public safety.”
Original Article: A cheaper, safer alternative to lithium-ion batteries: Aqueous rechargeable batteries
More from: Korea Institute of Science and Technology
The Latest Updates from Bing News
Go deeper with Bing News on:
Aqueous rechargeable batteries
- Breakthrough in Sodium Battery Chemistry Promises Lower Costs
A new mass synthesis process for sodium-containing sulfides could make all-solid-state sodium batteries more affordable and safer than lithium-ion batteries.
- Using sodium to develop rechargeable batteries may bolster the EU's green ambitions
A green industrial future for Europe may depend on an element that is part of a household staple: table salt. Dr. John Abou-Rjeily, a researcher at Tiamat Energy in France, is using sodium to develop ...
- Researchers develop high-energy-density aqueous battery based on halogen multi-electron transfer
Traditional non-aqueous lithium-ion batteries have a high energy density, but their safety is compromised due to the flammable organic electrolytes they utilize.
- New sodium battery that can be charged in seconds developed
R esearchers at the Korea Advanced Institute of Science and Technology (KAIST) have developed a high-power hybrid sodium-ion battery that can be charged in seconds.
Go deeper with Bing News on:
Energy storage systems
- Wendell Special Town Meeting to decide on battery storage bylaw
Residents are being summoned to Town Hall on Wednesday to have their voices heard on a bylaw proposed to regulate battery energy storage systems.A Special Town Meeting is slated to begin at 7 p.m., ...
- Battery costs have plummeted by 90% in less than 15 years, turbocharging renewable energy shift
The IEA's "Batteries and Secure Energy Transitions" report finds that capital costs for battery storage systems are projected to fall by up to 40 percent by ...
- RheEnergise to build first-of-a-kind hydro storage system
A demonstrator of the company’s long-duration hydro-energy storage system will be constructed at Sibelco’s Devon mine.
- Energy storage plant could power 124k homes
The storage system would be built off Foxhounds Lane in Southfleet, near Gravesend, and store enough energy to power more than 124,000 homes, planning documents say. EcoDev said the facility, covering ...
- The Novel Material Revolutionizing Energy Storage
Washington University in St. Louis scientists have developed a novel material that supercharges innovation in electrostatic energy storage, with the potential to revolutionize electronic devices.










