via AI.Nony.Mous
A new international study published in Nature Medicine and presented as a late-breaking abstract at the American Association of Neurological Surgeons (AANS) annual conference, shows great promise for patients with glioblastoma.
Drs. Farshad Nassiri and Gelareh Zadeh, neurosurgeons at the University Health Network (UHN) in Toronto, published the results of a Phase 1/2 clinical trial investigating the safety and effectiveness of a novel therapy which combines the injection of an oncolytic virus – a virus that targets and kills cancer cells – directly into the tumour, with intravenous immunotherapy.
The authors found that this novel combination therapy can eradicate the tumour in select patients, with evidence of prolonged survival.
Investigative work by the authors also revealed a new genetic signature within tumour samples that has the potential to predict which patients with glioblastoma are most likely to respond to treatment.
“The initial clinical trial results are promising,” says Dr. Zadeh, who is also Co-Director of the Krembil Brain Institute and a Senior Scientist at the Princess Margaret Cancer Centre. “We are cautiously optimistic about the long-term clinical benefits for patients.”
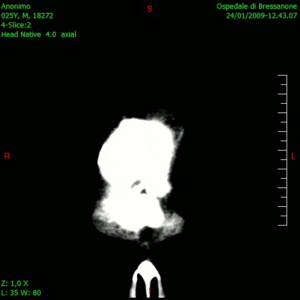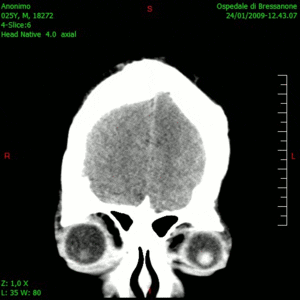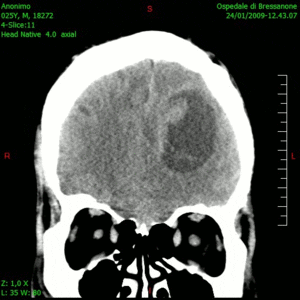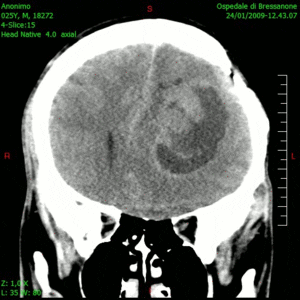
Glioblastoma is a notoriously difficult-to-treat primary brain cancer. Despite aggressive treatment, which typically involves surgical removal of the tumour and multiple chemotherapy drugs, the cancer often returns, at which point treatment options are limited.
Immune checkpoint inhibitors are effective treatments for a variety of cancers, but they have had limited success in treating recurrent glioblastoma. This novel therapy involves the combination of an oncolytic virus and immune checkpoint inhibition, using an anti-PD-1 antibody as a targeted immunotherapy.
First, the team delivered the virus by accurately localizing the tumour using stereotactic techniques and injecting the virus through a small hole and a purpose-built catheter. Then, patients received an anti-PD-1 antibody intravenously, every three weeks, starting one week after surgery.
“These drugs work by preventing cancer’s ability to evade the body’s natural immune response, so they have little benefit when the tumour is immunologically inactive – as is the case in glioblastoma,” explains Dr. Zadeh.
“Oncolytic viruses can overcome this limitation by creating a more favourable tumour microenvironment, which then helps to boost anti-tumour immune responses.”
The combination of the oncolytic virus and immune-checkpoint inhibition results in a “double hit” to tumours; the virus directly causes cancer cell death, but also stimulates local immune activity causing inflammation, leaving the cancer cells more vulnerable to targeted immunotherapy.
Dr. Zadeh and colleagues evaluated the innovative therapy in 49 patients with recurrent disease, from 15 hospital sites across North America.
UHN, which is the largest research and teaching hospital in Canada and the only Canadian institution involved in the study, treated the majority of the patients enrolled in the trial.
The results, published this week in Nature Medicine, show that this combination therapy is safe, well tolerated and prolongs patient survival. The therapy had no major unexpected adverse effects and yielded a median survival of 12.5 months – considerably longer than the six to eight months typically seen with existing therapies.
“We’re very encouraged by these results,” says Dr. Farshad Nassiri, first author of the study and a senior neurosurgery resident at the University of Toronto. “Over half of our patients achieved a clinical benefit – stable disease or better – and we saw some remarkable responses with tumours shrinking, and some even disappearing completely. Three patients remain alive at 45, 48 and 60 months after starting the clinical trial.”
“The findings of the study are particularly meaningful as the patients in the trial did not have tumour resection at recurrence – only injection of the virus – which is a novel treatment approach for glioblastoma. So, it’s really remarkable to see these responses,” says Dr. Zadeh.
“We believe the key to our success was delivering the virus directly into the tumour prior to using systemic immunotherapy. Our results clearly signal that this can be a safe and effective approach,” adds Dr. Nassiri.
The team also performed experiments to define mutations, gene expression, and immune features of each patient’s tumour. They discovered key immune features which could eventually help clinicians predict treatment responses and understand the mechanisms of glioblastoma resistance.
“In general, the drugs that are used in cancer treatment do not work for every patient, but we believe there is a sub-population of glioblastoma patients that will respond well to this treatment,” says Dr. Zadeh. “I believe this translational work, combining basic bench science and clinical trials, is key to moving personalized treatments for glioblastoma forward.”
This is one of the few clinical trials with favourable results for glioblastoma over the last decade, and it was truly a team effort.
“The trial would not have been possible without our incredible OR teams, research safety teams and researchers – including Dr. Warren Mason and his team at the Princess Margaret Cancer Centre – and our brave patients and their families. We’re also grateful to the Wilkins Family for providing the funds to enable us to complete trials that advance care for our patients,” says Dr. Zadeh.
The next steps for the group are to test the effectiveness of the combination therapy against other treatments in a randomized clinical trial.
“We are encouraged by these results, but there is still a lot of work ahead of us,” says Dr. Nassiri. “Our goal, as always, is to help our patients. That’s what motivates us to continue this research.”
Original Article: Study using novel approach for glioblastoma treatment shows promising results, extending survival
More from: University Health Network
The Latest Updates from Bing News
Go deeper with Bing News on:
Glioblastoma
- CAR T-Cell Therapy Could Be Adapted for Solid Tumors, Study Finds
One of the therapies—which used two novel components—proved superior in safely and effectively shrinking not only glioblastoma and pancreatic tumors but lung cancer tumors as well. CAR-T cell therapy, short for chimeric antigen receptor (CAR)-T cell ...
- Brain Cancer Canada awards The Dwayne Andrews Glioblastoma Grant for Research to a study on the impact of anesthetics on brain cancer proliferation
Brain Cancer Canada is excited to announce the issuance of an $80,000 grant to support groundbreaking research by Dr. Sujoy Banik and his team at Lawson Health Research Institute in London, Ontario, Canada.
- How Long Does Glioblastoma Take To Develop? A Review By Doctors
Expert opinion from Marcella Abunahman Pereira Specialization in Clinical Cardiology · 12 years of experience · Brazil Glioblastoma is a type of malignant tumor that affects the central nervous system.
- Ask A Doctor: How Long Does Glioblastoma Take To Develop?
Expert opinion from Marcella Abunahman Pereira Specialization in Clinical Cardiology · 12 years of experience · Brazil Glioblastoma is a type of malignant tumor that affects the central nervous system.
- Glioblastoma vaccine turns brain tumors from cold to hot in humans and dogs
A personalized mRNA-based cancer vaccine for glioblastoma with an “onion-like” delivery mechanism can prolong survival both in dogs and humans, the results of a small study have suggested.
Go deeper with Bing News on:
Oncolytic virus
- Oncolytic Viruses: A Promising New Option in Cancer Treatment
As part of its Speaking Out video series, Targeted Oncology spoke with Howard D. Edington, MD, about the benefits of using oncolytic viruses in cancer treatment.
- Oncolytic Virus Immunotherapy Market CAGR of 12.00%, is expected to Double Its Size by Upcoming Years
The promising outcomes during the early stage clinical trials with oncolytic virus immunotherapy is further expected to play an imperative tole in boosting the overall growth of the market. Also, the ...
- CG sets new bar in bladder cancer, validating rare biotech IPO success story
CG Oncology’s late-stage bladder cancer treatment notched a complete response in roughly three-quarters of treated patients, fueling optimism that the company’s Wall Street success story is no fluke.
- Oncolytic Virus to Be Studied in Advanced Lung Cancer
The oncolytic virus, MEM-288 will be studied in combination with Opdivo for patients with non-small cell lung cancer that progressed on standard therapy.
- Moffitt initiates groundbreaking clinical trial with oncolytic virus for non-small cell lung cancer
The innovative trial uses a novel oncolytic virus, MEM-288, in combination with the immune checkpoint inhibitor nivolumab. Spearheaded by Moffitt, in collaboration with Duke Cancer Institute ...