Researcher Dr. Jeffrey S. Weber led a clinical trial that found adding a vaccine to an immunotherapy reduced melanoma recurrence.
PHOTO: JULIANA THOMAS
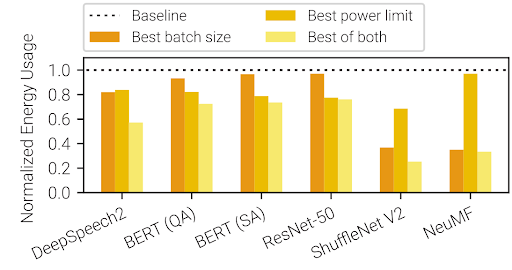
The combination of an experimental mRNA vaccine with an immunotherapy reduced the likelihood of melanoma recurring or causing death by 44 percent when compared to immunotherapy alone, a new clinical trial shows.
Led by researchers at NYU Langone Health and its Perlmutter Cancer Center, the randomized phase 2b trial involved men and women who had surgery to remove melanoma from lymph nodes or other organs and were at high risk of the disease returning in sites distant from the original cancer.
Among 107 study subjects who were injected with both the experimental vaccine, called mRNA-4157/V940, and the immunotherapy pembrolizumab, the cancer returned in 24 subjects (22.4 percent) within 3 years of follow-up, compared with 20 out of 50 (40 percent) who received only pembrolizumab.
“Our phase 2b study shows that a neoantigen mRNA vaccine, when used in combination with pembrolizumab, resulted in prolonged time without recurrence or death compared with pembrolizumab alone,” said study senior investigator Jeffrey S. Weber, MD, PhD, the deputy director of Perlmutter Cancer Center.
The phase 2b trial results are being presented at the annual meeting of the American Association for Cancer Research on April 16 in Orlando, Florida.
While randomized phase 3 trials test whether a treatment is superior to current standard therapies, phase 2 trials like the current study provide preliminary reassurance that one treatment is likely to be better than another, and lead to larger studies to confirm those results. Phase 3 trials of the combination of the mRNA-4157/V940 vaccine with pembrolizumab versus pembrolizumab alone are already planned at NYU Langone and a number of other medical centers globally, said Dr. Weber, the Laura and Isaac Perlmutter Professor of Oncology in the Department of Medicine at NYU Grossman School of Medicine.
Study results so far led the United States Food and Drug Administration (FDA) in February to grant Breakthrough Therapy Designation to mRNA-4157/V940 in combination with pembrolizumab, a designation designed to speed government reviews of trial results.
The current results highlight the role of immune system T cells capable of attacking viruses as well as cancers. To spare normal cells, this system uses “checkpoint” molecules on T cell surfaces to “turn off” their attack against viruses when they clear the infection. The body may recognize tumors as abnormal, but cancer cells hijack checkpoints to turn off, evade, and avoid immune responses. Immunotherapies like pembrolizumab seek to block checkpoints, making cancer cells more “visible” and vulnerable again to immune cells.
Immunotherapies have become the mainstay for treating melanoma, although they do not work for all patients because melanoma cells, known for their ability to evade the immune system, can become resistant to immunotherapy. For this reason, researchers have looked at adding vaccines. While most vaccines used today are designed to prevent infections, they can also be tailored to target proteins involved in cancer.
Like the COVID-19 vaccine, mRNA-4157/V940 is based on messenger RNA, a chemical cousin of DNA that provides instructions to cells for making proteins. mRNA cancer vaccines are designed to teach the body’s immune system to recognize cancer cells as different from normal cells. In designing a vaccine against melanoma, researchers attempted to trigger an immune response to specific abnormal proteins, called neoantigens, made by cancer cells.
Because the study volunteers all had their tumors removed, researchers were able to analyze their cells for neoantigens that were specific to each melanoma and create a personalized vaccine for each patient. As a result, T cells were produced specific to the neoantigen proteins encoded by the mRNA. Those T cells could then attack any melanoma cells trying to grow or spread.
Scientists involved in the study say that the personalized mRNA-4157/V940 vaccine took about 6 to 8 weeks to develop for each patient and could recognize as many as 34 neoantigens. Severe side effects were similar between the two arms of the study, they said, with fatigue being the most common side effect specific to the vaccine reported by patients.
Original Article: Adding New Vaccine Type to Leading Immunotherapy Dramatically Reduced Melanoma Recurrence
More from: NYU Langone Medical Center
The Latest Updates from Bing News
Go deeper with Bing News on:
Neoantigen mRNA vaccine
- MIT Technology Review
But the burst of renewed interest and activity around cancer vaccines is encouraging. And personalized vaccines might have a shot at succeeding where others have failed. The strategy makes sense for ...
- Moderna (MRNA) Q1 Earnings Beat Estimates, Revenues Down Y/Y
vaccine], mRNA-1010 (influenza vaccine), mRNA-4157/V940 [individualized neoantigen therapy (INT)] and mRNA-1083 (COVID-19 plus influenza combination vaccine). Data readouts on mRNA-1083 and mRNA ...
- Moderna plants new solid tumor trials for cancer vaccine, weeds PD-1 work after review
Much of Moderna’s focus is on nabbing FDA approval for a respiratory syncytial virus vaccine, a decision expected in a matter of weeks. | Much of Moderna’s focus is on nabbing FDA approval for a ...
- Moderna Reports First Quarter 2024 Financial Results and Provides Business Updates
Moderna's vaccine candidate for the prevention of norovirus (mRNA-1403) showed positive immunogenicity and safety data in a Phase 1 study. The Company is advancing toward a pivotal Phase 3 trial.
- Moderna Reports First Quarter 2024 Financial Results and Provides Business Updates
The PFS ready-to-use formulation will save pharmacists and clinicians time, potentially alleviating wait times and reducing the burden on pharmacy staff. In a study funded by Moderna, the PFS ...
Go deeper with Bing News on:
Melanoma
- What Are The Complications Melanoma? A Review By Doctors
Expert opinion from Ilya Aleksandrovskiy M.D., MBA · 5 years of experience · USA Common complications of melanoma include metastasis which is the most feared property of the cancer cells. It can ...
- UT Medical Center is first in Tennessee to offer newly FDA-approved melanoma treatment
The University of Tennessee Medical Center is currently the only hospital in the state to offer this type of therapy.
- Tips to determine if you might have melanoma
As the weather gets warmer you might be looking to soak up some sun, but you should also be wary of soaking up cancer.
- Melanoma in darker skin tones: Race and sex play a role, study finds
Melanoma, an aggressive form of skin cancer that accounts for 75% of all skin-cancer-related deaths, is often detected later in people with darker skin complexions—and the consequences can be ...
- How new immunotherapy is helping treat advanced stage 4 melanoma
Oncologists say the single-dose immunotherapy is currently approved for those with advanced stage 4 melanoma, who have not had success with other treatments. The process works by surgically removing a ...