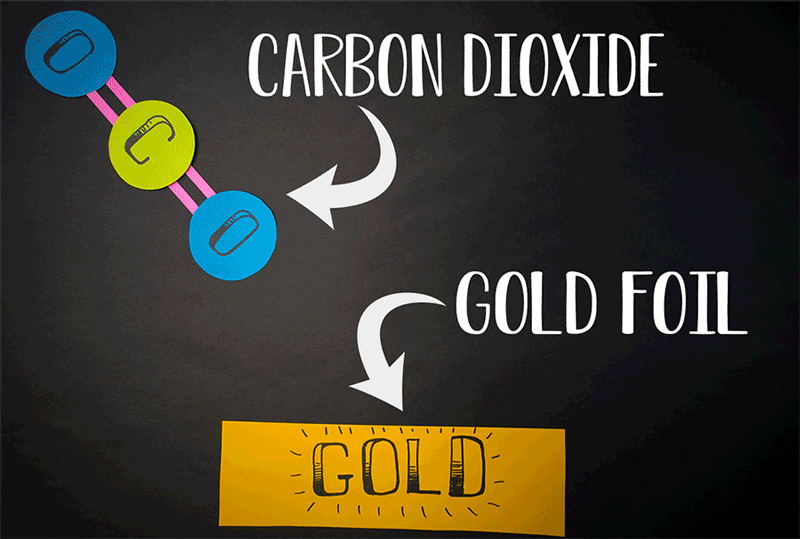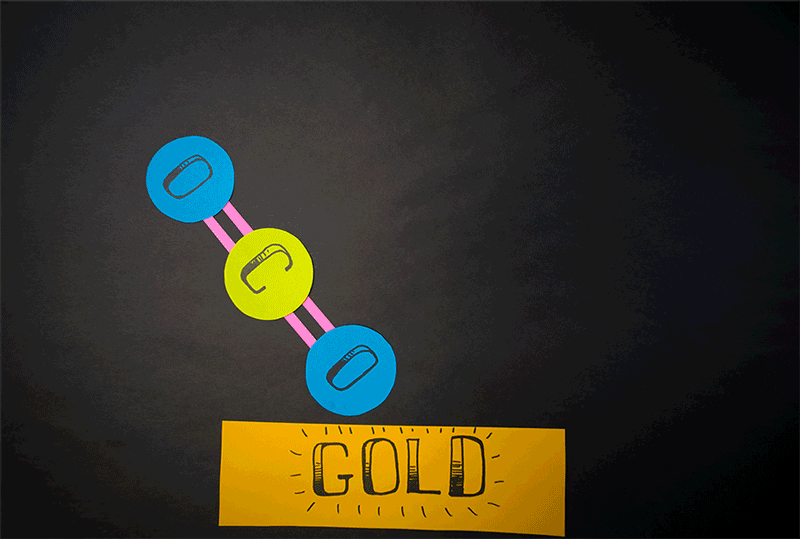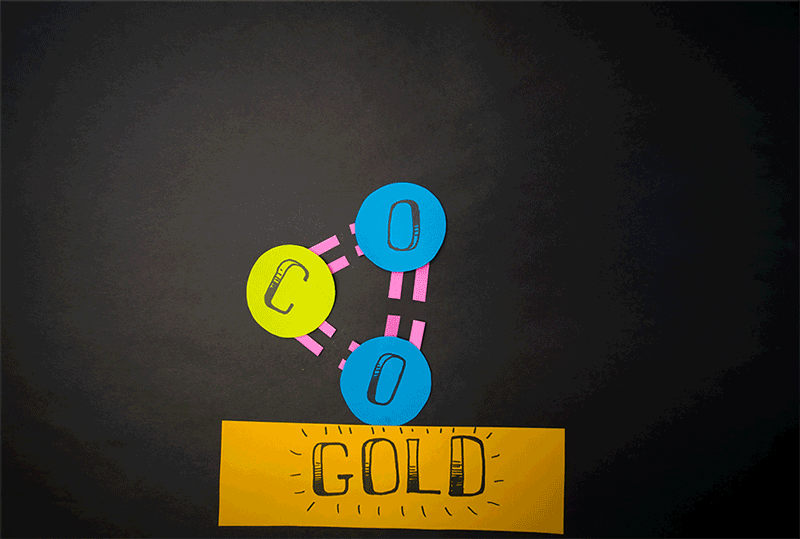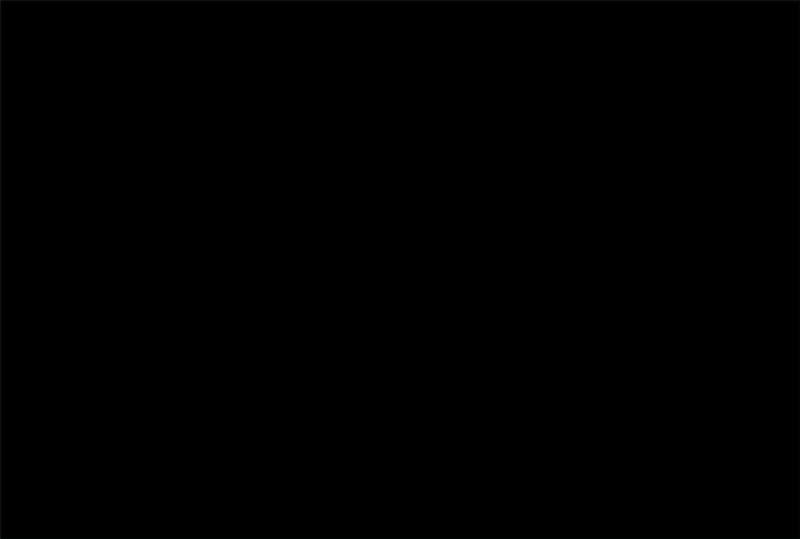
Credit: Caltech
Science fiction stories are chock full of terraforming schemes and oxygen generators for a very good reason—we humans need molecular oxygen (O2) to breathe, and space is essentially devoid of it. Even on other planets with thick atmospheres, O2 is hard to come by.
So, when we explore space, we need to bring our own oxygen supply. That is not ideal because a lot of energy is needed to hoist things into space atop a rocket, and once the supply runs out, it is gone.
One place molecular oxygen does appear outside of Earth is in the wisps of gas streaming off comets. The source of that oxygen remained a mystery until two years ago when Konstantinos P. Giapis, a professor of chemical engineering at Caltech, and his postdoctoral fellow Yunxi Yao, proposed the existence of a new chemical process that could account for its production. Giapis, along with Tom Miller, professor of chemistry, have now demonstrated a new reaction for generating oxygen that Giapis says could help humans explore the universe and perhaps even fight climate change at home. More fundamentally though, he says the reaction represents a new kind of chemistry discovered by studying comets.
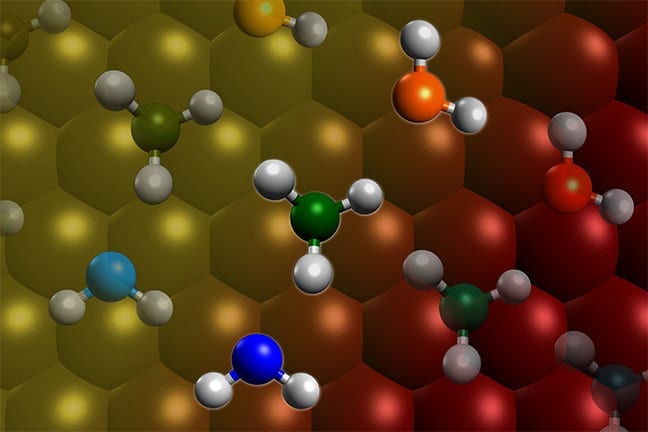
Most chemical reactions require energy, which is typically provided as heat. Giapis’s research shows that some unusual reactions can occur by providing kinetic energy. When water molecules are shot like extremely tiny bullets onto surfaces containing oxygen, such as sand or rust, the water molecule can rip off that oxygen to produce molecular oxygen. This reaction occurs on comets when water molecules vaporize from the surface and are then accelerated by the solar wind until they crash back into the comet at high speed.
Comets, however, also emit carbon dioxide (CO2). Giapis and Yao wanted to test if CO2could also produce molecular oxygen in collisions with the comet surface. When they found O2 in the stream of gases coming off the comet, they wanted to confirm that the reaction was similar to water’s reaction. They designed an experiment to crash CO2 onto the inert surface of gold foil, which cannot be oxidized and should not produce molecular oxygen. Nonetheless, O2 continued to be emitted from the gold surface. This meant that both atoms of oxygen come from the same CO2 molecule, effectively splitting it in an extraordinary manner.
“At the time we thought it would be impossible to combine the two oxygen atoms of a CO2molecule together because CO2 is a linear molecule, and you would have to bend the molecule severely for it to work,” Giapis says. “You’re doing something really drastic to the molecule.”
To understand the mechanism of how CO2 breaks down to molecular oxygen, Giapis approached Miller and his postdoctoral fellow Philip Shushkov, who designed computer simulations of the entire process. Understanding the reaction posed a significant challenge because of the possible formation of excited molecules. These molecules have so much energy that their constituent atoms vibrate and rotate around to an enormous degree. All that motion makes simulating the reaction in a computer more difficult because the atoms within the molecules move in complex ways.
“In general, excited molecules can lead to unusual chemistry, so we started with that,” Miller says. “But, to our surprise, the excited state did not create molecular oxygen. Instead, the molecule decomposed into other products. Ultimately, we found that a severely bent CO2can also form without exciting the molecule, and that could produce O2.”
The apparatus Giapis designed to perform the reaction works like a particle accelerator, turning the CO2 molecules into ions by giving them a charge and then accelerating them using an electric field, albeit at much lower energies than are found in a particle accelerator. However, he adds that such a device is not necessary for the reaction to occur.
“You could throw a stone with enough velocity at some CO2 and achieve the same thing,” he says. “It would need to be traveling about as fast as a comet or asteroid travels through space.”
That could explain the presence of small amounts of oxygen that have been observed high in the Martian atmosphere. There has been speculation that the oxygen is being generated by ultraviolet light from the sun striking CO2, but Giapis believes the oxygen is also generated by high-speed dust particles colliding with CO2 molecules.
He hopes that a variation of his reactor could be used to do the same thing at more useful scales—perhaps one day serving as a source of breathable air for astronauts on Mars or being used to combat climate change by pulling CO2, a greenhouse gas, out of Earth’s atmosphere and turning it into oxygen. He acknowledges, however, that both of those applications are a long way off because the current version of the reactor has a low yield, creating only one to two oxygen molecules for every 100 CO2 molecules shot through the accelerator.
“Is it a final device? No. Is it a device that can solve the problem with Mars? No. But it is a device that can do something that is very hard,” he says. “We are doing some crazy things with this reactor.”
Learn more: Comet Inspires Chemistry for Making Breathable Oxygen on Mars
The Latest on: New chemistry
[google_news title=”” keyword=”new chemistry” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: New chemistry
- Stand Atlantic Releases New Single And Music Video For ‘LOVE U ANWAY’ From Upcoming Album ‘Was Here’on May 9, 2024 at 11:33 pm
Aussie rock sensation Stand Atlantic has released a new single ‘LOVE U ANYWAY’ accompanied by a music video highlighting the band’s close bond. The new single includes Bonnie, Miki, Potter, and Jonno ...
- At the Met, Sleeping Beauty Wakes Up in the Chemistry Labon May 9, 2024 at 2:02 pm
The immersive show features fragile dresses inside airtight vitrines, overcoats growing grass, pat-’n-sniff walls and a hologram. Does it work?
- Catalyst search shows how computing can take the guesswork out of chemistryon May 8, 2024 at 7:51 am
Imagine synthesizing and then testing over 50 different complex molecules to identify the most effective catalyst for a particular chemical reaction. The traditional approach to developing new ...
- Polaroid makes "chemistry-loaded" upgrade to its B&W film to make it sharper, richer and unmatched in qualityon May 8, 2024 at 1:24 am
Polaroid has updated and improved the chemistry of its black-and-white film, elevating the instant photos able to be produced.
- A New Frontier in Chemistry: Roaming Reactions Shatter Old Assumptionson May 7, 2024 at 4:15 pm
Scientists have observed so-called 'roaming' chemical reactions, those that at certain points move away from the lowest minimum energy 'path of least resistance,' in highly excited energy states for ...
- Model Medicines' AI-Driven Drug Discovery Delivers Groundbreaking Advances in New Chemistry and Biologyon May 7, 2024 at 3:13 am
From helping doctors answer patient questions to making new drugs to cure hard diseases, there has been a lot of hype about how artificial intelligence (AI) is going to improve human health. In this ...
- Simulated chemistry: New AI platform designs tomorrow's cancer drugson May 6, 2024 at 8:48 am
Scientists at UC San Diego have developed a machine learning algorithm to simulate the time-consuming chemistry involved in the earliest phases of drug discovery, which could significantly streamline ...
- USC Football: Caleb Williams' Decorated New Bears Teammate Talks Developing Chemistryon May 5, 2024 at 6:39 pm
Former USC Trojans quarterback Caleb Williams is already making his presence known in the city of Chicago. He was drafted No. 1 overall by the Chicago Bears in the 2024 NFL Draft and has since been ...
- What instrumentation skills does industry want new chemists to have?on May 3, 2024 at 2:05 pm
Undergraduate chemists should learn how to use five key instruments to prepare for industry careers, a new study concludes. C&EN is published by ACS but is editorially independent.
- ‘Bad actors’ dominate new chemistry journal’s editorial board, critics allegeon April 29, 2024 at 5:00 pm
When François-Xavier Coudert came across a brand-new chemistry journal launched by the reputable publisher Taylor & Francis, he decided to check out the people who are running it. He recognized ...
via Bing News