UTMB develops a universal vaccine platform that’s cheaper and shelf stable
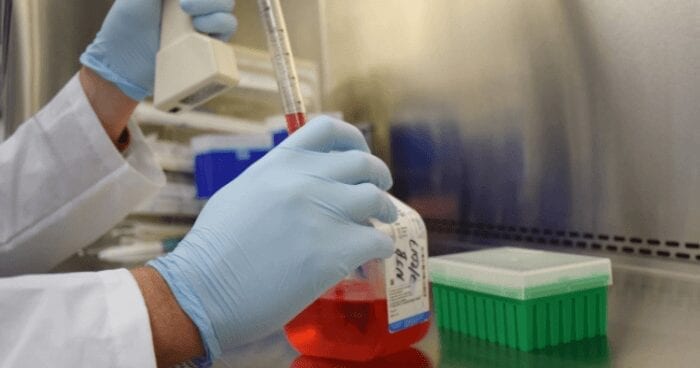
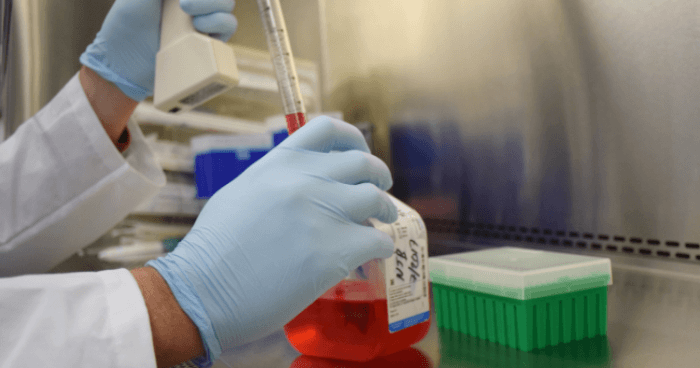
Researchers at The University of Texas Medical Branch at Galveston have developed a less expensive way to produce vaccines that cuts the cost of vaccine production and storage by 80 percent without decreasing safety or effectiveness. The findings are currently available in EBioMedicine.
Vaccines are the most effective way to prevent and eradicate infectious diseases. Currently, many vaccines have to be manufactured in cell culture or eggs, which is expensive and carries the risk of contaminations. In addition, most vaccines must be kept refrigerated during the transportation from manufacturers to health care clinics. In tropical and subtropical regions, such cold storage requirements could contribute to more than 80 percent of the vaccine cost.
“The ability to eliminate cell culture or eggs and cold storage will change the process of vaccine development,” said UTMB’s Pei-Yong Shi, professor in the department of biochemistry and molecular biology. “Importantly, this vaccine technology could potentially serve as a universal platform for development of live-attenuated vaccines for many viral pathogens.”
To achieve these goals, the UTMB team engineered a live-attenuated Zika vaccine in the DNA form. Once the DNA is delivered into our body, it launches the vaccine in our cells, leading to antibody production and other protective immunity. With this production method, there is no need to manufacture the vaccine in cell culture or eggs at factories. Because DNA molecules are shelf stable, the vaccine will not expire at warm temperatures and could be stockpiled at room temperature for years.
Using UTMB’s Zika vaccine as a model, the research group showed that the DNA platform worked very efficiently in mice. After a single low dose, the DNA vaccine protected mice from Zika virus infection, mother-to-fetus transmission during pregnancy and male reproductive tract infection and damage.
“This is the first study to demonstrate that, after a single low dose, a DNA vaccine could induce saturated protective immunity,” Shi said. “We will continue testing this promising Zika vaccine platform and then apply the platform to other viruses.”
Learn more: UTMB develops a universal vaccine platform that’s cheaper and shelf stable
The Latest on: Vaccine production
[google_news title=”” keyword=”vaccine production” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Vaccine production
- Leveraging vaccine potential to end meningitis scourgeon May 10, 2024 at 8:09 pm
Africa’s most populous country has a shallow domestic vaccine production system although its citizens suffer from many vaccine-preventable diseases. A major federal effort to revive vaccine production ...
- Bird Flu: U.S. Could Produce and Ship 100 Million Vaccine Doses Within Monthson May 10, 2024 at 9:45 am
By Shawn Radcliffe, Fact checked by Kevin Cyr, MD Unlike with COVID-19, the U.S. has candidate vaccines for the bird flu currently spreading in animals. Carlos Duarte/Getty Images The U.S. government ...
- Global Covid-19 Vaccine Development Tools Market Projected to Surge Forecasted to Exceed USD 51.6 Billion by 2032on May 9, 2024 at 7:17 pm
As the world continues to combat the global covid-19 vaccine development tools market has surged exponentially. According to recent projections, the global market for Covid-19 Vaccine Development ...
- A new cholera vaccine will increase supply, but will it be enough to manage global outbreaks?on May 9, 2024 at 1:31 am
Along with a new manufacturing plant that will be at full capacity next year, EuBiologics will be able to produce 50 million cholera vaccine doses in 2024, and 80 million in 2025, said Lynch — most of ...
- San Diego biotech news: Local maker of mRNA used in COVID-19 vaccine opens new facilityon May 8, 2024 at 10:27 pm
The local manufacturing facility will support the growing momentum behind mRNA-based medicines and vaccines beyond COVID-19.
- Vaccine Adjuvant from Tree Bark Has Been Produced by Engineered Yeaston May 8, 2024 at 12:01 pm
Researchers say new yeast biosynthetic platform provides "vast opportunities" to produce structural variants of QS-21 and identify new adjuvant leads.
- AstraZeneca Withdraws COVID-19 Vaccine Globally, Days After Side Effect Confession in UK Courton May 8, 2024 at 10:05 am
The company voluntarily revoked its marketing authorization and announced the cessation of vaccine production, rendering it unusable. This decision marks a significant development in the global ...
- An adjuvant made in yeast could lower vaccine cost and boost availabilityon May 8, 2024 at 9:57 am
Adjuvants make vaccines more effective, though one of the best is an expensive extract from the soap bark tree. To lower the cost and avoid the laborious extraction process from bark, synthetic ...
- AstraZeneca Withdraws Covid Vaccine After 3 Billion Shots Administered — But Why?on May 8, 2024 at 7:48 am
AstraZeneca discontinues widely used COVID-19 vaccine due to surplus and emerging variants, citing declining demand and emerging alternatives amid rare side effects reports.
via Bing News