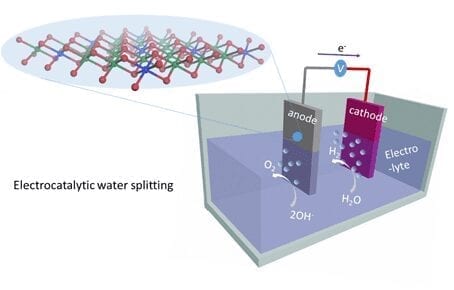
Solar-powered water splitting is a promising means of generating clean and storable energy. A novel catalyst based on semiconductor nanoparticles has now been shown to facilitate all the reactions needed for “artificial photosynthesis”.
In the light of global climate change, there is an urgent need to develop efficient ways of obtaining and storing power from renewable energy sources. The photocatalytic splitting of water into hydrogen fuel and oxygen provides a particularly attractive approach in this context. However, efficient implementation of this process, which mimics biological photosynthesis, is technically very challenging, since it involves a combination of processes that can interfere with each other. Now, LMU physicists led by Dr. Jacek Stolarczyk and Professor Jochen Feldmann, in collaboration with chemists at the University of Würzburg led by Professor Frank Würthner, have succeeded in demonstrating the complete splitting of water with the help of an all-in-one catalytic system for the first time. Their new study appears in the journal Nature Energy.
Technical methods for the photocatalytic splitting of water molecules use synthetic components to mimic the complex processes that take place during natural photosynthesis. In such systems, semiconductor nanoparticles that absorb light quanta (photons) can, in principle, serve as the photocatalysts. Absorption of a photon generates a negatively charged particle (an electron) and a positively charged species known as a ‘hole’, and the two must be spatially separated so that a water molecule can be reduced to hydrogen by the electron and oxidized by the hole to form oxygen. “If one only wants to generate hydrogen gas from water, the holes are usually removed rapidly by adding sacrificial chemical reagents,” says Stolarczyk. “But to achieve complete water splitting, the holes must be retained in the system to drive the slow process of water oxidation.” The problem lies in enabling the two half-reactions to take place simultaneously on a single particle – while ensuring that the oppositely charged species do not recombine. In addition, many semiconductors can be oxidized themselves, and thereby destroyed, by the positively charged holes.
Nanorods with spatially separated reaction sites
“We solved the problem by using nanorods made of the semiconducting material cadmium sulfate, and spatially separated the areas on which the oxidation and reduction reactions occurred on these nanocrystals,” Stolarczyk explains. The researchers decorated the tips of the nanorods with tiny particles of platinum, which act as acceptors for the electrons excited by the light absorption. As the LMU group had previously shown, this configuration provides an efficient photocatalyst for the reduction of water to hydrogen. The oxidation reaction, on the other hand, takes place on the sides of the nanorod. To this end, , the LMU researchers attached to the lateral surfaces a ruthenium-based oxidation catalyst developed by Würthner‘s team. The compound was equipped with functional groups that anchored it to the nanorod. “These groups provide for extremely fast transport of holes to the catalyst, which facilitates the efficient generation of oxygen and minimizes damage to the nanorods,” says Dr. Peter Frischmann, one of the initiators of the project in Würzburg.
The Latest on: Artificial photosynthesis
[google_news title=”” keyword=”artificial photosynthesis” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Artificial photosynthesis
- Toward artificial leaves that float on water, bubbling fuelon May 2, 2024 at 2:14 pm
Researchers have now made such an artificial leaf device using an organic photovoltaic (OPV) material. Depending on the catalyst used, the device either splits water to make hydrogen fuel or splits ...
- Hard Tech for Early-Stage Founders: HAX Invests in Startups Solving the Hardest Problems in Climate, Industrial Independence, and Healthcareon May 2, 2024 at 10:06 am
TechCrunch was proud to host HAX at Early Stage in Boston on April 25, 2024. Here’s an overview of their breakout session. There’s no tougher, early stage startup category than hard tech, and no one ...
- The 8 Best Grow Lights for Countertop Herbs, Small Gardens, and Helping Indoor Plants Thriveon April 30, 2024 at 2:33 pm
Narrow spectrum lights are noted for serious gardeners and plant nerds who need to support different stages of development. Blue-light spectrum (450-490 nm) grow lights are used for early-stage growth ...
- The Advantages of Going Solaron April 29, 2024 at 9:33 am
This article explores the numerous advantages of adopting solar energy, both from an environmental standpoint and its capacity to influence economic and personal development.
- Artificial Photosynthesis: A Game Changer for Clean Energyon March 20, 2024 at 7:13 pm
that brings us a step closer to the day carbon dioxide could be used as a resource.
- Is Artificial Photosynthesis the Next Source of Renewable Energy?on February 19, 2024 at 9:09 pm
Photosynthesis is the process by which plants convert sunlight into energy, inspiring sustainable energy solutions. Artificial photosynthesis aims to imitate the process of photosynthesis to ...
- The other half of the equationon October 20, 2023 at 12:43 am
Artificial photosynthesis — splitting water with light — is an attractive way to make hydrogen, but what happens to the oxygen? A catalyst that aids in the efficient production of gaseous ...
- Artificial photosynthesis machines invented to create oxygen in space for astronautson June 13, 2023 at 11:15 am
In a game-changing technological breakthrough, scientists are creating artificial photosynthesis devices that astronauts will use on future space missions. On our lush and abundant planet Earth ...
- Artificial Photosynthesis—Making Fuels Directly from Sunlighton May 13, 2022 at 5:40 pm
Nathan Lewis will present about the discovery and development of materials and a system architecture for the direct production of fuels from sunlight. The membrane-embedded microwire array approach is ...
via Bing News