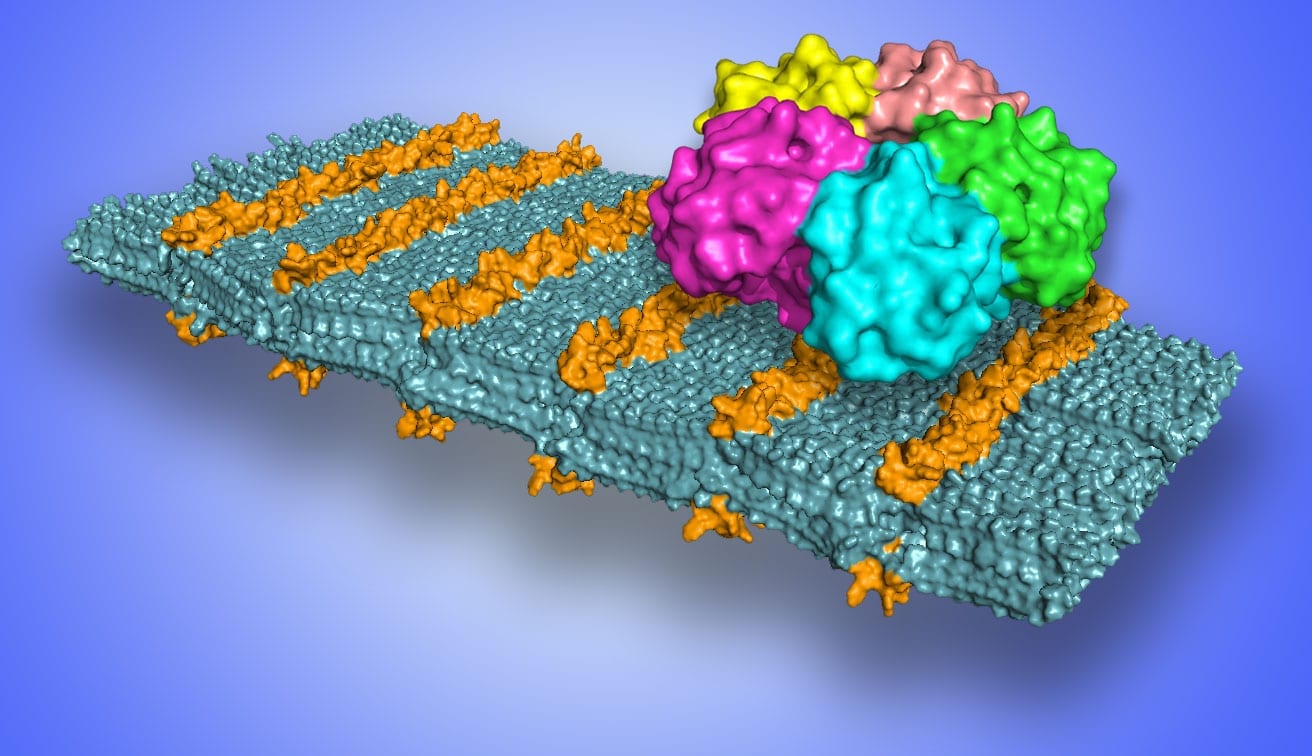
A Yale-led research team developed a new RNA therapy, delivered through the nose, to treat mice infected with West Nile Virus. The innovative approach reduced the virus in the brain, allowing the immune system to destroy the virus and develop long-term protection against West Nile Virus disease, the researchers said.
The findings, published in Cell Host & Microbe, may represent a breakthrough strategy for treating West Nile Virus after virus invasion of the brain and the central nervous system, noted senior author Priti Kumar, M.D., associate professor of infectious disease at Yale School of Medicine.
There are no approved vaccines or effective therapies for West Nile Virus disease, a mosquito-borne condition. While many infected individuals have no symptoms, others — particularly the very young and older adults — can develop severe neurological problems and even die from the disease. The sporadic nature of the disease makes it exceedingly difficult for testing and implementing vaccines, said Kumar.
To investigate a possible new therapy to treat West Nile Virus disease, Kumar and her colleagues focused on a small “interfering” RNA molecule developed in her lab. The RNA acts against multiple mosquito-borne flaviviruses. To direct the RNA to infected cells, they packaged it in a peptide derived from the rabies virus, which is able to enter nerve cells. The final step was to the deliver the therapy through the nose where it could bypass natural barriers protecting the brain.
With this novel approach, the researchers found that the therapy reduced the virus in the brain, preventing harm to nerve cells. The treated mice had a 90% survival rate a few days after infection compared to mice treated with placebo. Significantly, noted the researchers, in the surviving mice, the immune system cleared the virus throughout the body, and also enabled long-term protection against future exposure.
The researchers concluded that the treatment offers both a promising late-stage therapy and lifelong immunity. It “prevents pathology in the brain and gives the mice a chance to develop a robust immune response,” said Kumar.
While the anatomy of the murine nose differs from that of humans, the researchers plan to study the therapy further with the hope it will be broadly applicable. “In translation, it should be an effective strategy for people,” she said.
If that is the case, the intranasal RNA therapy could theoretically be developed for treatment of other mosquito-borne diseases, such as St. Louis encephalitis, Japanese encephalitis, and perhaps Zika, the researchers noted.
Learn more: Researchers develop a novel RNA-based therapy to target West Nile Virus
The Latest on: RNA therapy
[google_news title=”” keyword=”RNA therapy” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: RNA therapy
- OSU researchers develop device to improve delivery of gene therapy for lung diseaseon May 11, 2024 at 8:28 pm
Messenger RNA delivers instructions to cells for making a particular ... which triggers an immune response from the body. As a therapy for cystic fibrosis, the genetic material would fix the flaw in ...
- Buy Rating Affirmed for ProQR on Promising RNA Editing Technology and Solid Financial Footingon May 10, 2024 at 4:57 am
Chardan Capital analyst Keay Nakae has maintained their bullish stance on PRQR stock, giving a Buy rating today. Keay Nakae has given his Buy ...
- US FDA grants breakthrough therapy designation to Avidity Biosciences’ delpacibart etedesiran to treat myotonic dystrophy type 1on May 10, 2024 at 1:30 am
US FDA grants breakthrough therapy designation to Avidity Biosciences’ delpacibart etedesiran to treat myotonic dystrophy type 1: San Diego Friday, May 10, 2024, 14:00 Hrs [IST] ...
- RNA-LPAs Induce Immune Responses in Glioblastoma Patientson May 9, 2024 at 7:45 am
RNA lipid particle aggregates (RNA-LPAs) induced immune responses against glioblastoma in a first-in-human trial, according to a report published in Cell.1 The RNA-LPAs were designed to “enhance the ...
- FDA grants breakthrough status for myotonic dystrophy type 1 RNA therapyon May 8, 2024 at 9:20 am
Today, the US Food and Drug Administration (FDA) has granted Breakthrough Therapy designation (BTD) to delpacibart etedesiran (AOC 1001). This is USA-based Avidity Biosciences’ (Nasdaq: RNA) lead ...
- RNA blood test may predict antidepressant responseon May 8, 2024 at 3:57 am
The analysis by US biotech Circular Genomics focuses on an assay that looks for a circular RNA (circRNA) blood biomarker that the company says can predict response to sertraline, a commonly prescribed ...
- Avidity Biosciences Receives FDA Breakthrough Therapy Designation for Delpacibart Etedesiran (AOC 1001) for Treatment of Myotonic Dystrophy Type 1on May 8, 2024 at 3:00 am
Avidity Biosciences, Inc. (Nasdaq: RNA), a biopharmaceutical company committed to delivering a new class of RNA therapeutics called Antibody Oligonucleotide Conjugates (AOCs™), today announced that ...
- Avidity Receives Breakthrough Therapy Designation For Its Myotonic Dystrophy Type 1 Drug Candidateon May 8, 2024 at 12:00 am
(RTTNews) - Avidity Biosciences, Inc. (RNA) Wednesday said the Food and Drug Administration (FDA) has granted Breakthrough Therapy designation to delpacibart etedesiran (AOC 1001), the company's lead ...
- Nanoparticle researchers develop microfluidic platform for better delivery of gene therapy for lung diseaseon May 7, 2024 at 7:14 am
Drug delivery researchers at Oregon State University have developed a device with the potential to improve gene therapy for patients with inherited lung diseases such as cystic fibrosis.
via Bing News