Courtesy of the researchers
MIT researchers discover astonishing behavior of water confined in carbon nanotubes.
It’s a well-known fact that water, at sea level, starts to boil at a temperature of 212 degrees Fahrenheit, or 100 degrees Celsius. And scientists have long observed that when water is confined in very small spaces, its boiling and freezing points can change a bit, usually dropping by around 10 C or so.
But now, a team at MIT has found a completely unexpected set of changes: Inside the tiniest of spaces — in carbon nanotubes whose inner dimensions are not much bigger than a few water molecules — water can freeze solid even at high temperatures that would normally set it boiling.
“All bets are off when you get really small,” Strano says. “It’s really an unexplored space.”
The discovery illustrates how even very familiar materials can drastically change their behavior when trapped inside structures measured in nanometers, or billionths of a meter. And the finding might lead to new applications — such as, essentially, ice-filled wires — that take advantage of the unique electrical and thermal properties of ice while remaining stable at room temperature.
The results are being reported today in the journal Nature Nanotechnology, in a paper by Michael Strano, the Carbon P. Dubbs Professor in Chemical Engineering at MIT; postdoc Kumar Agrawal; and three others.
“If you confine a fluid to a nanocavity, you can actually distort its phase behavior,” Strano says, referring to how and when the substance changes between solid, liquid, and gas phases. Such effects were expected, but the enormous magnitude of the change, and its direction (raising rather than lowering the freezing point), were a complete surprise: In one of the team’s tests, the water solidified at a temperature of 105 C or more. (The exact temperature is hard to determine, but 105 C was considered the minimum value in this test; the actual temperature could have been as high as 151 C.)
“The effect is much greater than anyone had anticipated,” Strano says.
It turns out that the way water’s behavior changes inside the tiny carbon nanotubes — structures the shape of a soda straw, made entirely of carbon atoms but only a few nanometers in diameter — depends crucially on the exact diameter of the tubes. “These are really the smallest pipes you could think of,” Strano says. In the experiments, the nanotubes were left open at both ends, with reservoirs of water at each opening.
Even the difference between nanotubes 1.05 nanometers and 1.06 nanometers across made a difference of tens of degrees in the apparent freezing point, the researchers found. Such extreme differences were completely unexpected. “All bets are off when you get really small,” Strano says. “It’s really an unexplored space.”
In earlier efforts to understand how water and other fluids would behave when confined to such small spaces, “there were some simulations that showed really contradictory results,” he says. Part of the reason for that is many teams weren’t able to measure the exact sizes of their carbon nanotubes so precisely, not realizing that such small differences could produce such different outcomes.
In fact, it’s surprising that water even enters into these tiny tubes in the first place, Strano says: Carbon nanotubes are thought to be hydrophobic, or water-repelling, so water molecules should have a hard time getting inside. The fact that they do gain entry remains a bit of a mystery, he says.
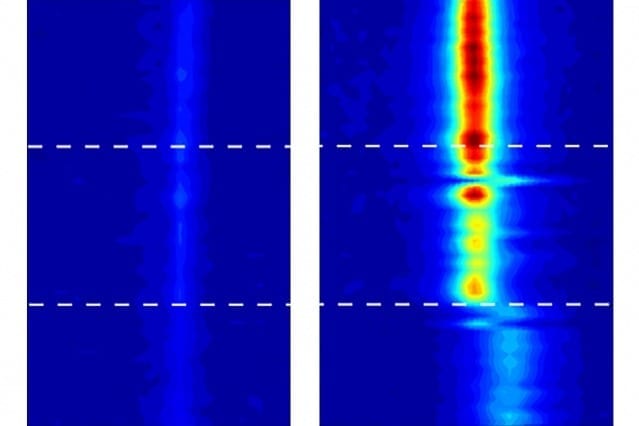
Strano and his team used highly sensitive imaging systems, using a technique called vibrational spectroscopy, that could track the movement of water inside the nanotubes, thus making its behavior subject to detailed measurement for the first time.
The team can detect not only the presence of water in the tube, but also its phase, he says: “We can tell if it’s vapor or liquid, and we can tell if it’s in a stiff phase.” While the water definitely goes into a solid phase, the team avoids calling it “ice” because that term implies a certain kind of crystalline structure, which they haven’t yet been able to show conclusively exists in these confined spaces. “It’s not necessarily ice, but it’s an ice-like phase,” Strano says.
Because this solid water doesn’t melt until well above the normal boiling point of water, it should remain perfectly stable indefinitely under room-temperature conditions. That makes it potentially a useful material for a variety of possible applications, he says. For example, it should be possible to make “ice wires” that would be among the best carriers known for protons, because water conducts protons at least 10 times more readily than typical conductive materials. “This gives us very stable water wires, at room temperature,” he says.
Learn more: Inside tiny tubes, water turns solid when it should be boiling
The Latest on: Water wires
[google_news title=”” keyword=”Water wires” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Water wires
- Charleston Water Taxi Puts Yamaha Outboards to the Teston May 20, 2024 at 5:00 am
Yamaha Marine’s Blue Life series today launched a new feature spotlighting the Charleston Water Taxi (link), a signature low country form of transportation for many who travel the Ashley and Cooper ...
- Segway Navimow i110N review: A boundary-wire-free bot for lesson May 20, 2024 at 3:05 am
At a glance Expert's Rating Pros ・Best navigation technology in a sub-$1,000 robot mower ・No line-of-sight requirement from the mower to the GPS antenna ・Outstanding cutting performance ・Handles ...
- Tech firm creates purification system which makes canal water drinkableon May 20, 2024 at 1:52 am
Technology company IF created a miniature vapour compression distillation system without consumable parts or chemicals, which works in 45 minutes to begin purifying previously untreatable water – and ...
- State Police Arrest Pair for Copper Wire Theft from New Castle Area Construction Siteon May 19, 2024 at 12:36 pm
Delaware State Police arrested David Mitchell, 55, of Newark, Delaware, and Colleen Floyd, 32, of Delaware City, Delaware, for theft of copper wires and related offenses from a New Castle area The ...
- Newton hunts for lead pipes in town water system. What homeowners need to knowon May 17, 2024 at 1:36 am
The 129-year-old water system may include hundreds of lead service pipes. But how to find them without digging up the town?
- The Best Water Shoeson May 16, 2024 at 11:47 pm
Also, we’ve noticed stock issues with the Crocs Men’s Swiftwater Mesh Wave shoes, which we mention in Other good water shoes. A slippery rock shouldn’t spoil a white-water rafting adventure ...
- English village told to boil water after outbreak from parasite sickens more than 45on May 16, 2024 at 6:11 pm
A scenic fishing village in southwestern England is on its third day under instructions to boil its tap water after a parasite sickened more than 45 people LONDON -- A scenic fishing village in ...
- Cape Hatteras National Seashore Visitors Cautioned About Exposed Wires, Pipes, Septic Systemson May 16, 2024 at 8:59 am
Utilities and septic systems exposed by the Atlantic Ocean at Cape Hatteras National Seashore in North Carolina have prompted an advisory to visitors to the seashore.
- Water main break damages Pawtucket roadon May 15, 2024 at 10:32 am
Police were dispatched to Central Avenue late on Tuesday night for a report of a water main break at American Insulated Wire.
via Bing News