While the human race will always leave its carbon footprint on the Earth, it must continue to find ways to lessen the impact of its fossil fuel consumption.
“Carbon capture” technologies – chemically trapping carbon dioxide before it is released into the atmosphere – is one approach. In a recent study, Cornell University researchers disclose a novel method for capturing the greenhouse gas and converting it to a useful product – while producing electrical energy.
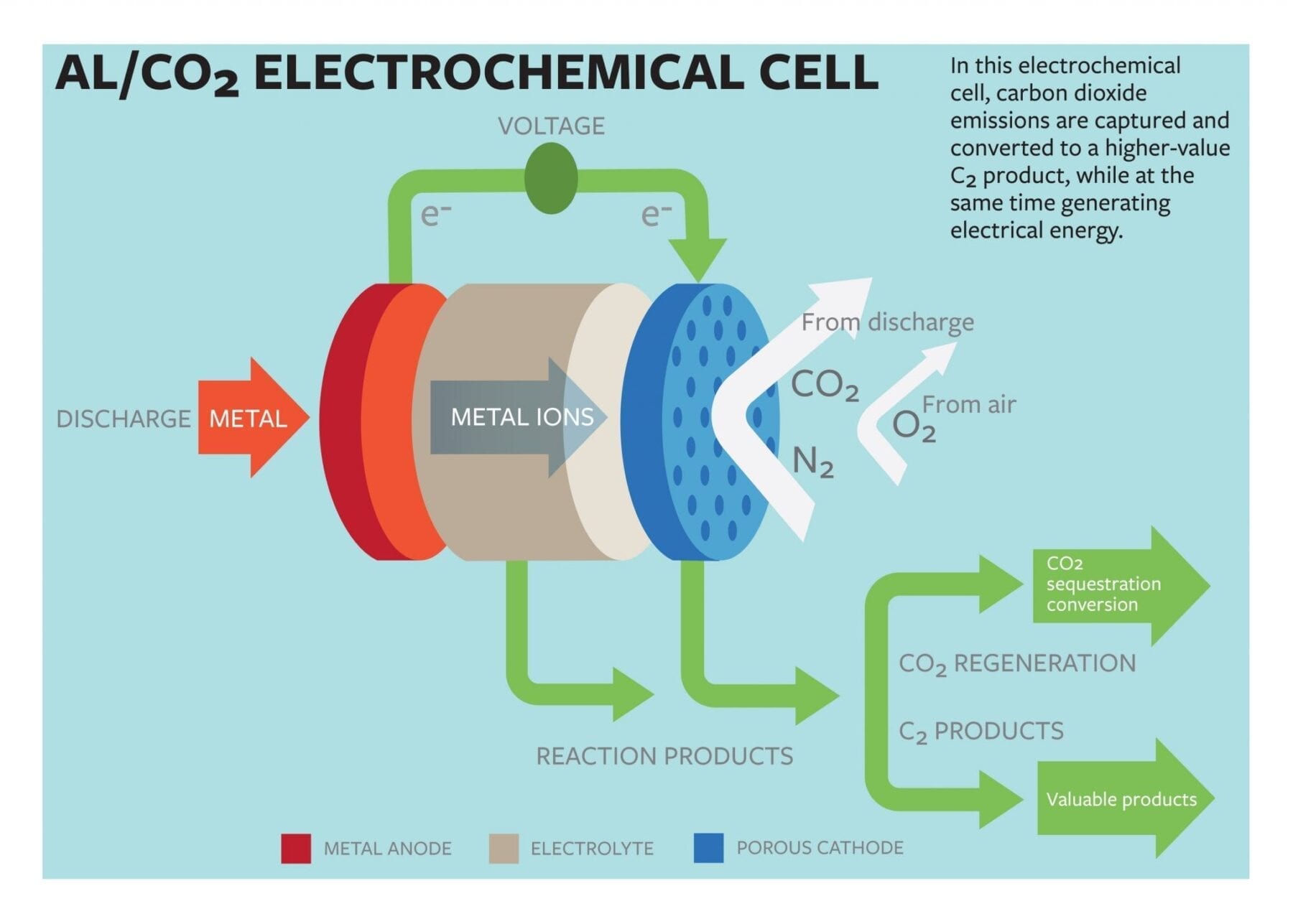
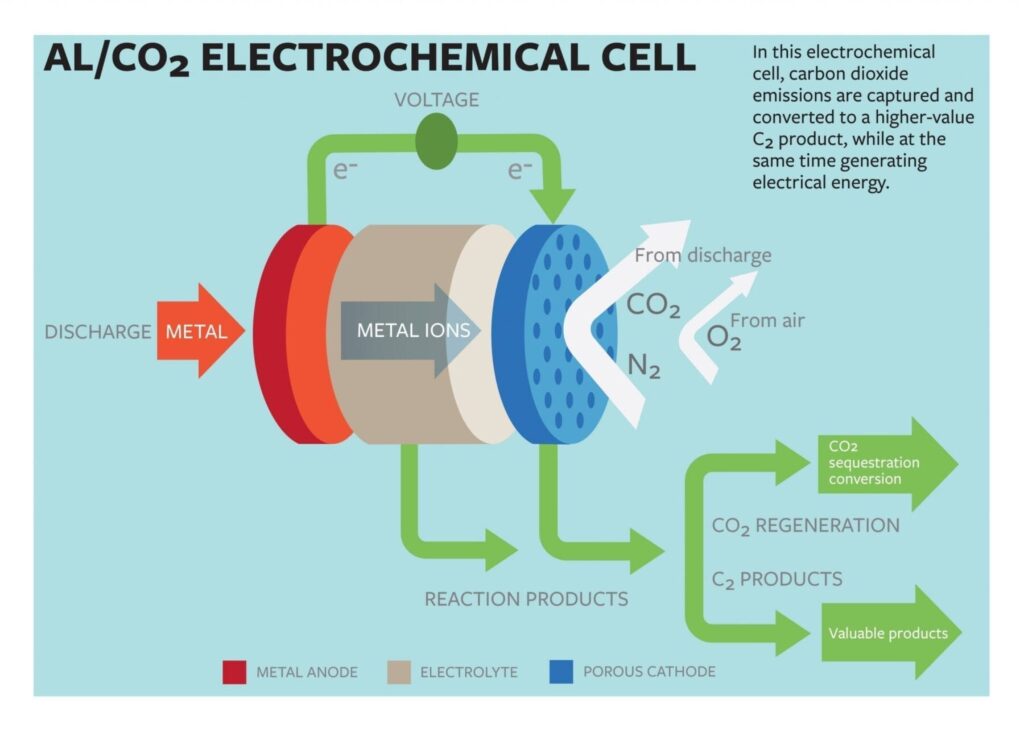
Lynden Archer, the James A. Friend Family Distinguished Professor of Engineering, and doctoral student Wajdi Al Sadat have developed an oxygen-assisted aluminum/carbon dioxide power cell that uses electrochemical reactions to both sequester the carbon dioxide and produce electricity.
Their paper, “The O2-assisted Al/CO2 electrochemical cell: A system for CO2capture/conversion and electric power generation,” was published July 20 in Science Advances.
The group’s proposed cell would use aluminum as the anode and mixed streams of carbon dioxide and oxygen as the active ingredients of the cathode. The electrochemical reactions between the anode and the cathode would sequester the carbon dioxide into carbon-rich compounds while also producing electricity and a valuable oxalate as a byproduct.
In most current carbon-capture models, the carbon is captured in fluids or solids, which are then heated or depressurized to release the carbon dioxide. The concentrated gas must then be compressed and transported to industries able to reuse it, or sequestered underground. The findings in the study represent a possible paradigm shift, Archer said.
“The fact that we’ve designed a carbon capture technology that also generates electricity is, in and of itself, important,” he said. “One of the roadblocks to adopting current carbon dioxide capture technology in electric power plants is that the regeneration of the fluids used for capturing carbon dioxide utilize as much as 25 percent of the energy output of the plant. This seriously limits commercial viability of such technology. Additionally, the captured carbon dioxide must be transported to sites where it can be sequestered or reused, which requires new infrastructure.”
The group reported that their electrochemical cell generated 13 ampere hours per gram of porous carbon (as the cathode) at a discharge potential of around 1.4 volts. The energy produced by the cell is comparable to that produced by the highest energy-density battery systems.
Another key aspect of their findings, Archer says, is in the generation of superoxide intermediates, which are formed when the dioxide is reduced at the cathode. The superoxide reacts with the normally inert carbon dioxide, forming a carbon-carbon oxalate that is widely used in many industries, including pharmaceutical, fiber and metal smelting.
“A process able to convert carbon dioxide into a more reactive molecule such as an oxalate that contains two carbons opens up a cascade of reaction processes that can be used to synthesize a variety of products,” Archer said, noting that the configuration of the electrochemical cell will be dependent on the product one chooses to make from the oxalate.
Al Sadat, who worked on onboard carbon capture vehicles at Saudi Aramco, said this technology in not limited to power-plant applications. “It fits really well with onboard capture in vehicles,” he said, “especially if you think of an internal combustion engine and an auxiliary system that relies on electrical power.”
He said aluminum is the perfect anode for this cell, as it is plentiful, safer than other high-energy density metals and lower in cost than other potential materials (lithium, sodium) while having comparable energy density to lithium. He added that many aluminum plants are already incorporating some sort of power-generation facility into their operations, so this technology could assist in both power generation and reducing carbon emissions.
A current drawback of this technology is that the electrolyte – the liquid connecting the anode to the cathode – is extremely sensitive to water. Ongoing work is addressing the performance of electrochemical systems and the use of electrolytes that are less water-sensitive.
Learn more: Cornell scientists convert carbon dioxide, create electricity
The Latest on: Electrochemical cell
[google_news title=”” keyword=”Electrochemical cell” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Electrochemical cell
- This invention converts alcohol into hydrogen: this is how you can have the most futuristic fuelon May 17, 2024 at 5:00 pm
The mechanism of power cells is electrochemical, and not combustion. This allows them to be noisy while off the grid and emitting zero pollution particles. Methanol is one of the preferred renewable ...
- Hydrogen Vehicle market is projected to grow at a CAGR of 22.7% by 2034: Visiongainon May 17, 2024 at 9:24 am
Visiongain has published a new report entitled Hydrogen Vehicle Market Report 2024-2034: Forecasts by Technology Type (Fuel Cell Electric Vehicles (FCEVs), Hydrogen Internal Combustion Engine Vehicles ...
- Battery Cell Characterization: Electrochemical and Prismatic Cellson May 17, 2024 at 8:11 am
This article explores battery cell characterizing, including coin, pouch, electrochemical, and prismatic cells.
- New Carbon-Capture Batteries Store Renewable Energy, Help Climateon May 17, 2024 at 1:53 am
Researchers at the Department of Energy’s Oak Ridge National Laboratory are developing battery technologies to fight climate change in two ways, by expanding the use of renewable energy and capturing ...
- Researchers use new X-ray technique to explore affordable alternative fuel cell materials: 'This is critical'on May 16, 2024 at 3:30 am
"Experiments like this help bring fuel cell researchers closer to an ideal catalyst." Researchers use new X-ray technique to explore affordable alternative fuel cell materials: 'This is critical' ...
- Carbon-capture batteries developed to store renewable energy, help climateon May 15, 2024 at 2:02 pm
Researchers at the Department of Energy’s Oak Ridge National Laboratory are developing battery technologies to fight climate change in two ways, by expanding the use of renewable energy and capturing ...
- Fuel cells inspire new lead-carbon battery technologyon May 15, 2024 at 1:50 pm
PEM fuel cell architecture (Proton Exchange Membrane) inspires battery developers to innovative integration of lead-carbon batteries with an ...
- Energy harvester powers remote devices in the IoTon May 9, 2024 at 6:04 am
The pyroelectrochemical cell converts thermal energy into electricity and stores it. Source: Brian Maffly, University of Utah ...
- 1 Reason Goldman Sachs Analysts Are Skeptical of Plug Power Stockon May 9, 2024 at 4:18 am
This brings us to Goldman Sachs and its recent research note on Plug Power stock. The firm's forecast claims that investors will remain skeptical of the longest-duration pockets of the equity market.
- Exploring Electrochemical Ozone Production at the Molecular Levelon May 6, 2024 at 4:59 pm
They are studying how electrochemical ozone production (EOP) works at the molecular level to develop a better catalyst that is efficient, economical, and long-lasting. The study was published in the ...
via Bing News