New technologies are starved for efficient and inexpensive catalysts. The best materials are made up of nanoparticles, whose properties are the result of their small size. The single catalyst particles have, however, an ugly tendency to cluster into larger particles, thereby reducing their effectiveness.
A group of scientists from the International School of Advanced Studies in Trieste and the DEMOCRITOS centre of the Istituto Officina dei Materiali of the Italian National Research Council (IOM-CNR), with the collaboration of other institutions, have developed a material that maintains the stability of a “dispersed” catalyst, thus maximising the efficiency of the process and decreasing costs and wastage. The study has just been published in Nature Communications.
Platinum is one of the costly metals used as catalysts in new technologies employed for industrial chemical processes, renewable energy sources, pollution control and many other purposes. In particular, it is used for fuel cells, devices that turn chemical energy directly into electrical energy, without combustion. Research has shown that the greatest efficiency is achieved when the catalyst is available in the form of nanoparticles (smaller than 10-9 m). Simply put, the greater the dispersion of the material and the smaller the size of the particles, the more is it available for catalysis. Unfortunately, the laws of thermodynamics cause the particles to “stick” to one another and form larger clusters, which is why the material becomes less effective over time. So what can be done to maintain maximal dispersion of the “nanopowder”?
A group of SISSA/CNR IOM scientists (with the collaboration of the Univerzita Karlova in Prague) has studied a way to produce tiny platinum grains consisting of one atom only and to keep them dispersed in a stable manner, by exploiting the properties of the substrate on which they rest.
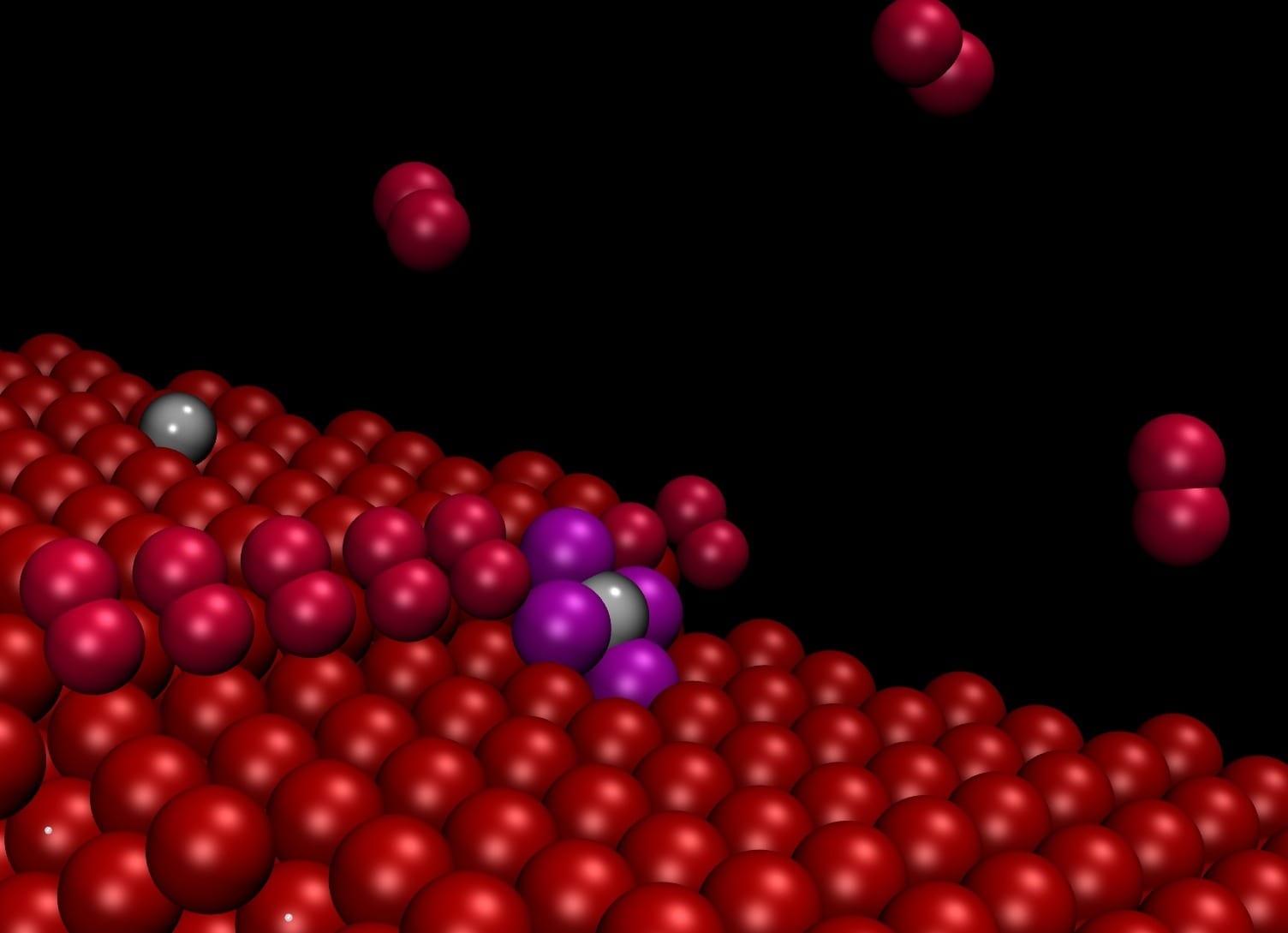
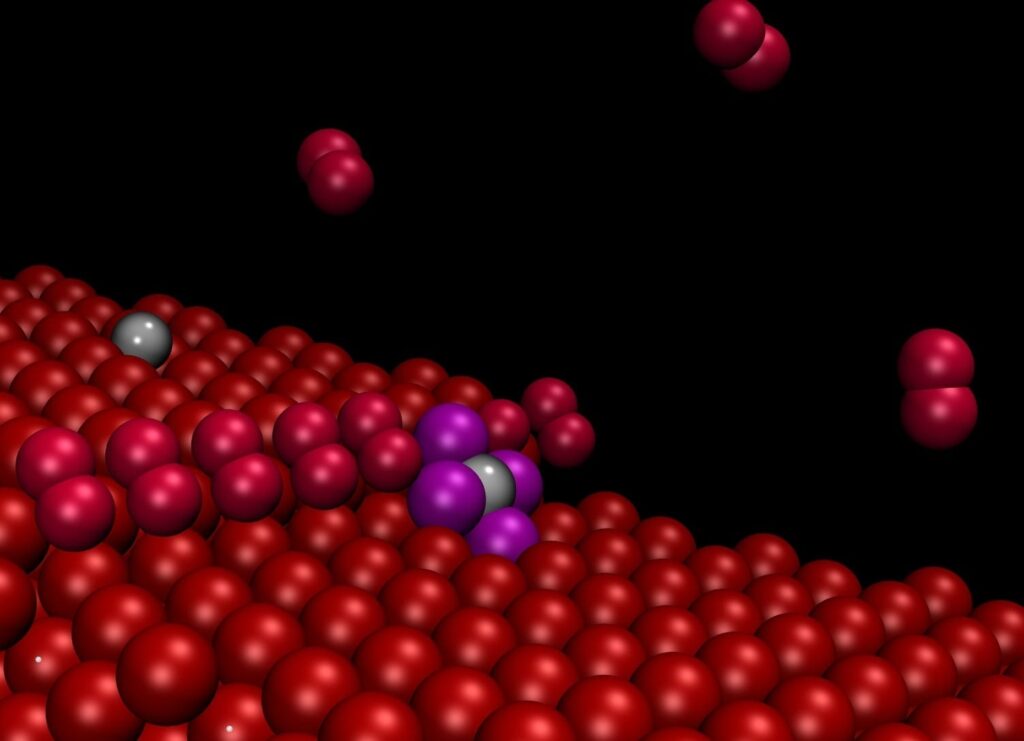
“Theoretical work demonstrated that irregularities in the surface known as steps and observed in experiments conducted at the Trieste Synchrotron tend to attract and separate the nanoparticles, causing them to remain literally attached in the form of single atoms”, explains Stefano Fabris, CNR-IOM/SISSA research fellow.
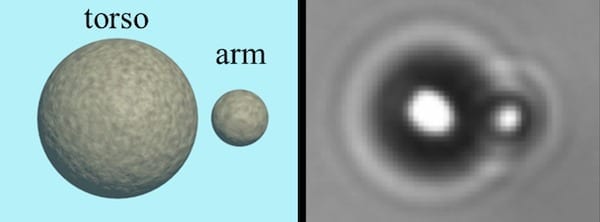
“The particles adhering to the steps were no longer visible even using an atomic resolution microscope” explains Nguyen-Dung Tran, a SISSA PhD student. “However, their presence was detected by spectroscopy, so they were indeed there, but they were no longer visible or free to move around”. “Our computer simulations solved this dilemma, showing that the particles on the steps are reduced to single atoms” adds Matteo Farnesi Camellone (CNR-IOM), another author of the study.
“If the surface is engineered to contain a large number of these defects, then the force that binds the particles to the substrate effectively offsets the aggregation force”, explains Fabris. The theoretical work, led by Fabris, allowed the researchers to develop a “system model” on the computer able predict the behaviour of the material. The model’s predictions were confirmed by the experimental measurements. Materials like this can be used for fuel cell electrodes, with far lower costs than the current ones.
“Reducing the amount of platinum used in fuel cell electrodes is a priority, not only to contain costs but also to ensure environmental sustainability, as also indicated by the recent European directives” concludes Fabris. The European project ChipCAT, which funded this research, aims precisely to achieve this goal.
Learn more: Nanoparticles on nanosteps
The Latest on: Dispersed catalysts
[google_news title=”” keyword=”dispersed catalysts” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Dispersed catalysts
- Exploring Electrochemical Ozone Production at the Molecular Levelon May 6, 2024 at 4:59 pm
These nickel atoms, now dispersed in the solution near the catalyst, can catalyze chemical reactions that ultimately lead to ozone generation. If we want to make a better electrocatalyst, we need to ...
- Team describes MXene-supported PtCo bimetallic catalyst for hydrogen evolution in acidic conditionson May 6, 2024 at 12:51 pm
Hydrogen energy is considered a promising solution with high energy density and zero pollution emissions. Currently, hydrogen is mainly derived from fossil fuels, which increases energy consumption ...
- Health Catalyston May 6, 2024 at 10:51 am
When Health Catalyst begins working with a hospital system, it makes a sweet offer: Move your engineers over to the Utah-based software firm, and they’ll keep your data running and analyzable it ...
- MXene-Supported PtCo Catalysts for Hydrogen Evolutionon May 6, 2024 at 10:42 am
In a recent paper published in Frontiers in Energy, researchers from Beijing University of Technology developed a new hydrogen production catalyst using MXene material and a small amount of platinum ...
- Hydrogen energy advances with efficient low-cost catalysts on MXeneon May 5, 2024 at 5:00 pm
A research group of Kai-Ling Zhou, Yang Yang, Yuhong Jin, Hao Wang from Beijing University of Technology and Institute of Deep-Sea Science and Engineering, Chinese Academy of Sciences fabricate small ...
- MXene supported PtCo bimetallic catalyst for hydrogen evolution in acidic conditionson May 4, 2024 at 5:00 pm
A research group of Kai-Ling Zhou, Yang Yang, Yuhong Jin, Hao Wang from Beijing University of Technology and Institute of Deep-Sea Science and Engineering, Chinese Academy of Sciences fabricate small ...
- Adequately stabilized and exposed Cu/CuₓO heterojunction on porous carbon nanofiberson April 28, 2024 at 5:00 pm
Inexpensive and readily available copper-based catalysts are considered ideal ... In this research, highly-dispersed copper nanoparticles within carbon nanofiber were firstly prepared via ...
- Catalyst synthesis articles from across Nature Portfolioon April 26, 2024 at 5:00 pm
Catalyst synthesis is the process of fabricating ... facilitated by the rapid transformation of monomeric dispersed metal oxides. The in situ generation of reactive (di)gallenes from a gallium ...
- Magnetism boosts hydrogen production in model catalystson April 15, 2024 at 5:00 pm
In the search for green hydrogen, the design of efficient catalyst materials that increase the efficiency and speed of the chemical reaction that produces (green) hydrogen is essential.
- Murata’s Groundbreaking Ceramic Catalyst Material Eliminates Precious Metal Reliance and Cuts Carbon Emissionson April 10, 2024 at 5:00 pm
Alternatively, ceramic catalyst materials have active elements dispersed in their crystal structure and do not degrade even in high-temperature operation, leading to an extended lifespan compared to ...
via Bing News