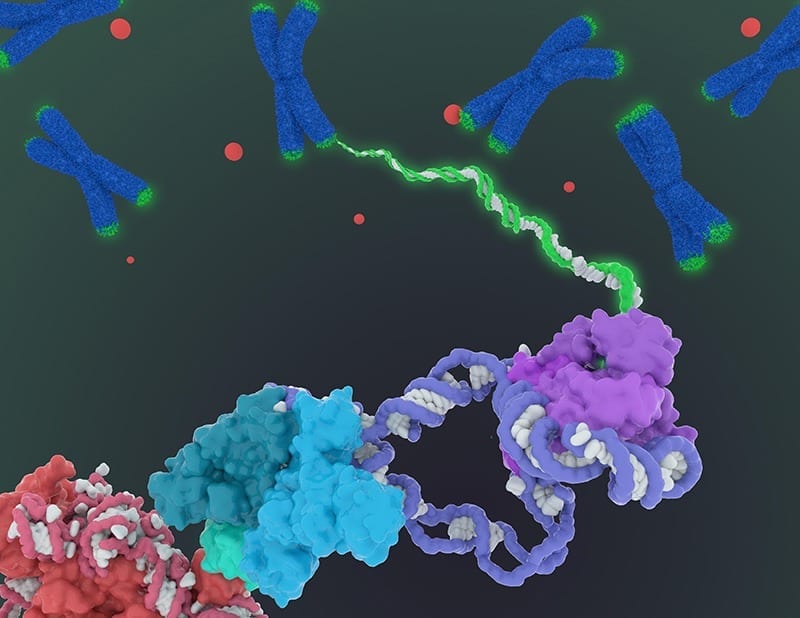
A new procedure can quickly and efficiently increase the length of human telomeres, the protective caps on the ends of chromosomes that are linked to aging and disease, according to scientists
Treated cells behave as if they are much younger than untreated cells, multiplying with abandon in the laboratory dish rather than stagnating or dying.
The procedure, which involves the use of a modified type of RNA, will improve the ability of researchers to generate large numbers of cells for study or drug development, the scientists say. Skin cells with telomeres lengthened by the procedure were able to divide up to 40 more times than untreated cells. The research may point to new ways to treat diseases caused by shortened telomeres.
Telomeres are the protective caps on the ends of the strands of DNA called chromosomes, which house our genomes. In young humans, telomeres are about 8,000-10,000 nucleotides long. They shorten with each cell division, however, and when they reach a critical length the cell stops dividing or dies. This internal “clock” makes it difficult to keep most cells growing in a laboratory for more than a few cell doublings.
‘Turning back the internal clock’
“Now we have found a way to lengthen human telomeres by as much as 1,000 nucleotides, turning back the internal clock in these cells by the equivalent of many years of human life,” said Helen Blau, PhD, professor of microbiology and immunology at Stanford and director of the university’s Baxter Laboratory for Stem Cell Biology. “This greatly increases the number of cells available for studies such as drug testing or disease modeling.”
A paper describing the research was published today in the FASEB Journal. Blau, who also holds the Donald E. and Delia B. Baxter Professorship, is the senior author. Postdoctoral scholar John Ramunas, PhD, of Stanford shares lead authorship with Eduard Yakubov, PhD, of the Houston Methodist Research Institute.
The researchers used modified messenger RNA to extend the telomeres. RNA carries instructions from genes in the DNA to the cell’s protein-making factories. The RNA used in this experiment contained the coding sequence for TERT, the active component of a naturally occurring enzyme called telomerase. Telomerase is expressed by stem cells, including those that give rise to sperm and egg cells, to ensure that the telomeres of these cells stay in tip-top shape for the next generation. Most other types of cells, however, express very low levels of telomerase.
Transient effect an advantage
The newly developed technique has an important advantage over other potential methods: It’s temporary. The modified RNA is designed to reduce the cell’s immune response to the treatment and allow the TERT-encoding message to stick around a bit longer than an unmodified message would. But it dissipates and is gone within about 48 hours. After that time, the newly lengthened telomeres begin to progressively shorten again with each cell division.
The transient effect is somewhat like tapping the gas pedal in one of a fleet of cars coasting slowly to a stop. The car with the extra surge of energy will go farther than its peers, but it will still come to an eventual halt when its forward momentum is spent. On a biological level, this means the treated cells don’t go on to divide indefinitely, which would make them too dangerous to use as a potential therapy in humans because of the risk of cancer.
The researchers found that as few as three applications of the modified RNA over a period of a few days could significantly increase the length of the telomeres in cultured human muscle and skin cells. A 1,000-nucleotide addition represents a more than 10 percent increase in the length of the telomeres. These cells divided many more times in the culture dish than did untreated cells: about 28 more times for the skin cells, and about three more times for the muscle cells.
“We were surprised and pleased that modified TERT mRNA worked, because TERT is highly regulated and must bind to another component of telomerase,” said Ramunas. “Previous attempts to deliver mRNA-encoding TERT caused an immune response against telomerase, which could be deleterious. In contrast, our technique is nonimmunogenic. Existing transient methods of extending telomeres act slowly, whereas our method acts over just a few days to reverse telomere shortening that occurs over more than a decade of normal aging. This suggests that a treatment using our method could be brief and infrequent.”
Read more: Telomere extension turns back aging clock in cultured human cells, study finds
The Latest on: Telomere extension
[google_news title=”” keyword=”Telomere extension” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Telomere extension
- Could cutting down calories influence longevity?on April 24, 2024 at 5:00 pm
Share on Pinterest Caloric restriction may influence aging by reducing telomere shortening. Image credit: Aleksandr Zubkov/Getty Images. Animal studies suggest that restricting calories may ...
- ILF3 safeguards telomeres from aberrant homologous recombination as a telomeric R-loop readeron April 22, 2024 at 11:18 am
Dysregulated R-loops can cause stalled replication forks and telomere instability. However, how R-loops are recognized and regulated, is still not well understood, particularly at telomeres.
- Findings suggest ILF3 may function as a reader of telomeric R-loops to help maintain telomere homeostasison April 21, 2024 at 5:00 pm
Dysregulated R-loops can cause stalled replication forks and telomere instability. However, how R-loops are recognized and regulated, is still not well understood, particularly at telomeres.
- What Flexion and Extension Means for Your Workoutson April 21, 2024 at 5:00 pm
Two terms that come up often when it comes to movement are flexion and extension. Understanding concepts like flexion versus extension can help you better understand the purpose of your workouts ...
- Keeping Telomeres in Their Placeson April 21, 2024 at 5:00 pm
Cells use homologous recombination to stitch double-stranded breaks (DSB) back together and the enzyme telomerase to cap exposed ends of a DNA strand with a repetitive DNA sequence called a telomere.
- Book Review: A New Chapter in the Quest for a Longer Lifeon April 19, 2024 at 12:53 am
In “Why We Die,” biologist and Nobel laureate Venki Ramakrishnan explores the science of aging and life extension.
- Telomeres and Telomerases in Canceron April 13, 2024 at 5:00 pm
Some cells that do not produce telomerase are still able to maintain telomere length. Roger Reddel, [5] of the Children's Medical Research Institute in Sydney, Australia, is investigating this ...
- Haven't filed your taxes yet? Here's how to get an extension from the IRS.on April 12, 2024 at 11:06 am
If you're one of the millions of Americans who have procrastinated filing their taxes ahead of the April 15 deadline, there is a way to get some breathing room: File for an extension. The step ...
- How to get a last-minute tax filing extensionon April 11, 2024 at 7:05 pm
An extension affords filers more time but comes with limitations. Tax season, beloved by few and dreaded by many, comes to a close on Monday. For some tardy filers, though, the task will just be ...
- Epithalon Peptide Research: Sleep, Aging, Tumors, and Moreon April 11, 2024 at 4:18 am
and promoting telomere extension to potentially achieve anti-aging outcomes within cells. This professional advice has been compiled to address readers' inquiries about the properties and ...
via Bing News