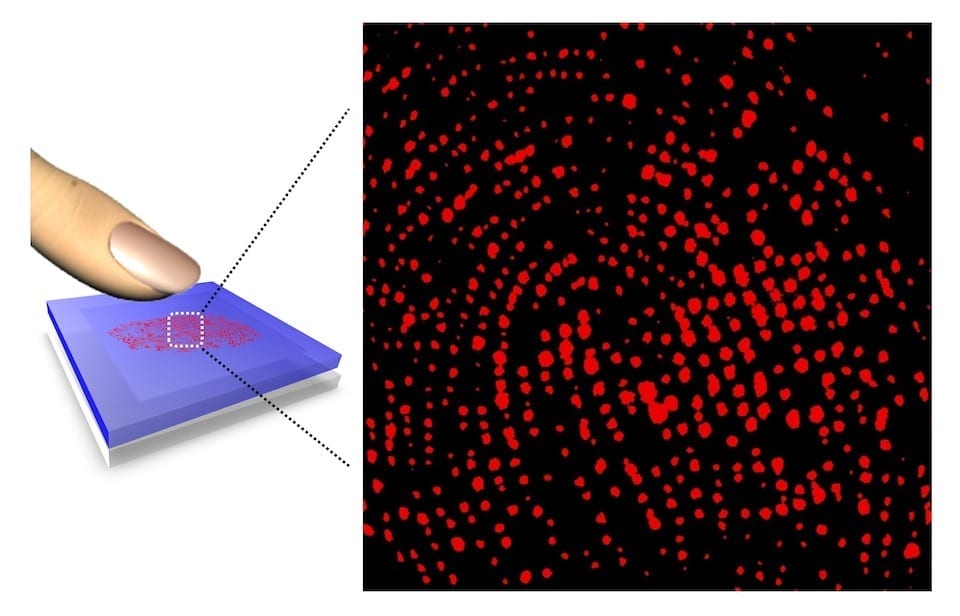
Material that could change electronics industry is shown to be very mobile in water and likely to cause negative environmental impacts if spilled
In a first-of-its-kind study of how a material some think could transform the electronics industry moves in water, researchers at the University of California, Riverside Bourns College of Engineering found graphene oxide nanoparticles are very mobile in lakes or streams and therefore likely to cause negative environmental impacts if released.
Graphene oxide nanoparticles are an oxidized form of graphene, a single layer of carbon atoms prized for its strength, conductivity and flexibility. Applications for graphene include everything from cell phones and tablet computers to biomedical devices and solar panels.
The use of graphene and other carbon-based nanomaterials, such as carbon nanotubes, are growing rapidly. At the same time, recent studies have suggested graphene oxide may be toxic to humans.
As production of these nanomaterials increase, it is important for regulators, such as the Environmental Protection Agency, to understand their potential environmental impacts, said Jacob D. Lanphere, a UC Riverside graduate student who co-authored a just-published paper about graphene oxide nanoparticles transport in ground and surface water environments.
“The situation today is similar to where we were with chemicals and pharmaceuticals 30 years ago,” Lanphere said. “We just don’t know much about what happens when these engineered nanomaterials get into the ground or water. So we have to be proactive so we have the data available to promote sustainable applications of this technology in the future.”
The paper co-authored by Lanphere, “Stability and Transport of Graphene Oxide Nanoparticles in Groundwater and Surface Water,” was published in a special issue of the journal Environmental Engineering Science.
Other authors were: Sharon L. Walker, an associate professor and the John Babbage Chair in Environmental Engineering at UC Riverside; Brandon Rogers and Corey Luth, both undergraduate students working in Walker’s lab; and Carl H. Bolster, a research hydrologist with the U.S. Department of Agriculture in Bowling Green, Ky.
Walker’s lab is one of only a few in the country studying the environmental impact of graphene oxide. The research that led to the Environmental Engineering Science paper focused on understanding graphene oxide nanoparticles’ stability, or how well they hold together, and movement in groundwater versus surface water.
The researchers found significant differences.
In groundwater, which typically has a higher degree of hardness and a lower concentration of natural organic matter, the graphene oxide nanoparticles tended to become less stable and eventually settle out or be removed in subsurface environments.
In surface waters, where there is more organic material and less hardness, the nanoparticles remained stable and moved farther, especially in the subsurface layers of the water bodies.
The researchers also found that graphene oxide nanoparticles, despite being nearly flat, as opposed to spherical, like many other engineered nanoparticles, follow the same theories of stability and transport.
The Latest on: Graphene
[google_news title=”” keyword=”Graphene” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Graphene
- Scientists create black arsenic visible infrared photodetectorson May 8, 2024 at 11:44 am
Examples of such crystals include graphene, black phosphorus (BP), and transition metal dichalcogenides (TMDs). With their atomic thickness, high carrier mobility, and tunable bandgaps, ...
- Finnlines RO/RO and RoPax fleets to get XGIT-FUEL hull coatingson May 8, 2024 at 11:29 am
FUEL, an innovative graphene-based hard foul release hull coating, across its RO/RO and RoPax fleets Starting with a first vessel in 2022, Finnlines has already applied the high-performance hull ...
- Finnlines rolls out fuel and emissions reducing hull coatingon May 8, 2024 at 9:41 am
Finnlines is to roll out a graphene-based hard foul release hull coating across its ro-ro and ro-pax fleets following a seven per cent reduction in fuel consumption and emissions on four of its ...
- Wonder Material 'More Remarkable' Than Graphene Has Medical Potentialon May 7, 2024 at 8:55 am
Borophene is already thinner and more conductive than graphene, and scientists have altered it to make it even more special.
- Graphene Manufacturing Group Announces Closing of $3.47M Marketed Offering, Including Exercise In Full of the Over-Allotment Optionon May 7, 2024 at 6:13 am
NOT FOR DISTRIBUTION TO UNITED STATES NEWSWIRE SERVICES OR FOR DISSEMINATION IN THE UNITED STATES BRISBANE, Australia, May 07, 2024 (GLOBE NEWSWIRE) -- (TSX-V: GMG) (“GMG” or the “Company”) is pleased ...
- 'Better than graphene' material development may improve implantable technologyon May 6, 2024 at 7:01 am
Move over, graphene. There's a new, improved two-dimensional material in the lab. Borophene, the atomically thin version of boron first synthesized in 2015, is more conductive, thinner, lighter, ...
- Charge up to six devices simultaneously with this successfully Indiegogo-funded power bank, further on saleon May 2, 2024 at 6:00 am
Charge your gadgets using the Flash Pro Plus 100W USB-C 25000mAh Graphene Power Bank, now further on sale for $199.97.
- Wigner crystal appears in bilayer grapheneon May 2, 2024 at 1:30 am
Researchers at Princeton University in the US say they have made the first direct observation of a Wigner crystal – a structure consisting solely of electrons arranged in a lattice-like configuration.
- Graphene at 20: why the ‘wonder material’ is finally coming goodon April 30, 2024 at 6:31 am
Strong, light and with amazing electronic properties, graphene has always been touted as the “wonder material”. But two decades after it was first isolated, James McKenzie believes the graphene is ...
- Beyond Graphene: A New World of 2D Materials Is Opening Upon April 28, 2024 at 9:07 pm
Materials that are incredibly thin, only a few atoms thick, exhibit unique properties that make them appealing for energy storage, catalysis, and water purification. Researchers at Linköping ...
via Bing News