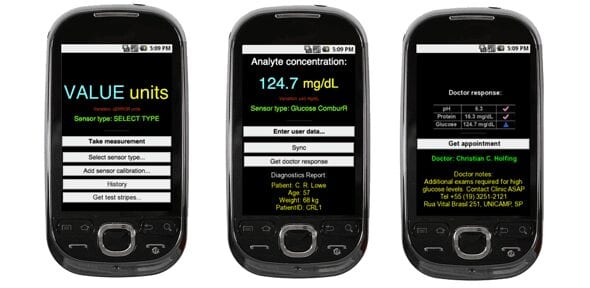
Results of a clinical trial of Cologuard show unprecedented rates of precancer and cancer detection by a noninvasive test.
The detection rates are similar to those reported for colonoscopy. The results were published in the March 20 issue of the New England Journal of Medicine (NEJM). Cologuard was co-developed by Mayo Clinic and Exact Sciences. Cologuard, is a noninvasive sDNA test for the early detection of colorectal precancer and cancer. The Cologuard test is based on a stool sample that is analyzed for DNA signatures of precancer or cancer. The samples are easily collected, mailed from home, requires no bowel preparation, medication restriction or diet change. The clinical trial, called the DeeP-C study, included 10,000 patients and was designed to determine how well Cologuard detects precancer and cancer.
The study also compared Cologuard to the fecal immunochemical test for occult blood (FIT). The study was conducted at 90 medical centers throughout the United States and Canada. “Cologuard detection rates of early stage cancer and high-risk precancerous polyps validated in this large study were outstanding and have not been achieved by other noninvasive approaches,” says the study’s author David Ahlquist M.D., a Mayo Clinic gastroenterologist and co-inventor of the Cologuard test. “It is our hope that this accurate and user-friendly test will expand screening effectiveness and help curb colorectal cancer rates in much the same way as regular Pap smear screening has done for cervical cancer.” In the study, all patients received Cologuard, FIT and colonoscopy. Colonoscopy was the reference method. Major findings reported in the study include:
- Sensitivity of Cologuard for cancer was 92 percent overall, and 94 percent for the earliest and most curable cancer stages (stages I and II).
- Sensitivity was 69 percent for precancerous polyps at greatest risk to progress to cancer (i.e., those containing high-grade dysplasia).
- Cologuard detected significantly more cancers and significantly more precancerous polyps than did FIT.
“The most important finding of the study is the high sensitivity of Cologuard for curable stage colorectal cancer, which represents the highest sensitivity of any noninvasive test to date,” says Thomas Imperiale, M.D., a Professor of Medicine at Indiana University Medical Center in Indianapolis and a study author. “It is also significant to note that these results were achieved in a robustly conducted multicenter study.”
Colorectal cancer is often considered the most preventable, yet least prevented, cancer. Nearly 50 percent of adults age 50 and older have not been screened as recommended and, as a result, colorectal cancer has become the second-leading cause of cancer death in the United States. Colorectal cancer is highly treatable if found early, and it is preventable if the precancerous polyps at greatest risk of progressing can be detected. “Dr. Ahlquist’s work exemplifies our goal at Mayo Clinic, which is the relentless pursuit of innovation aimed to help our patients. This research will transform how we think about colorectal cancer screening going forward,” says Vijay Shah, M.D., chair of Mayo Clinic’s Division of Gastroenterology and Hepatology. Cologuard works by testing a patient’s stool for altered DNA shed during digestion. Altered DNA is known to occur within colorectal cancers and precancerous polyps.
The test also examines the stool for the presence of blood, another possible indicator of colorectal cancer. Combining the data from the stool DNA test and the blood test into a single result provides a comprehensive, powerful screening approach, which is reflected in the study results. Because of its accessibility and ease of use, researchers hope the test will increase the number of people who will choose to be screened for colorectal cancer. Screening is important because, if cancer is detected early, removing polyps during a colonoscopy can prevent the cancer. “This test has the potential to bring colon cancer screening to many patients who might otherwise go without any screening,” says Kenneth Wang, M.D., a Mayo Clinic gastroenterologist, director of the Advanced Endoscopy Group and president of the American Society for Gastrointestinal Endoscopy. “I’m hopeful that this will increase the number of patients obtaining lifesaving colonoscopy with early detection and removal of precancerous and early cancerous polyps.”
The Latest on: Colorectal Cancer Screening
[google_news title=”” keyword=”Colorectal Cancer Screening” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Colorectal Cancer Screening
- What lifestyle factors may contribute to an increased risk of colorectal cancer in youngsters?on May 17, 2024 at 11:13 am
Piles (haemorrhoids) or Anaemia in individuals over 40 may indicate colorectal cancer and require further investigation through colonoscopy. Screening is recommended for individuals over 50, with at ...
- Offering both colonoscopy and at-home tests to study patients doubled colorectal cancer screeningon May 16, 2024 at 1:26 pm
The rate of colorectal cancer screenings more than doubled when patients were given a choice between which type of screening they wanted—a take-home kit or colonoscopy—compared to those who were only ...
- Olympus to Highlight EVIS X1 Endoscopy System and Colorectal Cancer Screening Solutions at Digestive Disease Week Annual Meetingon May 16, 2024 at 11:15 am
Olympus, a global medical technology company committed to making people's lives healthier, safer and more fulfilling, announced today that it will highlight its EVIS X1™ endoscopy system and ...
- Beloved Chicago Reporter Mike Lowe, 44, Reveals Stage 3 Colon Cancer Diagnosis: 'Never Die Easy'on May 16, 2024 at 9:54 am
Lowe added that “I’ll be motivated by Walter Payton’s motto ‘Never Die Easy’” and shared a photo of himself reading the like-titled autobiography of the Chicago Bears running back, who died of bile ...
- Offering both colonoscopy and at-home tests doubled colorectal cancer screeningon May 15, 2024 at 5:00 pm
In a trial analyzing how messages were framed in an underserved population, offering colorectal cancer screening options resulted in the highest screening rate ...
- Lung cancer is the deadliest of all cancers. Screening could save lives, if more people had accesson May 14, 2024 at 12:35 pm
Many medical organizations have been recommending lung cancer screening for decades for those at high risk of developing the disease. But in 2022, less than 6% of people in the U.S. eligible for ...
- Guardant Health preps for FDA advisory panel review of its blood-based colorectal cancer screening teston May 13, 2024 at 6:48 am
The USPSTF last updated its colorectal cancer screening guidelines in 2021, saying all adults between the ages of 45 and 75 should get checked out, including with a stool test each year or a ...
- Colorectal cancer cases have increased in kids over the last 2 decades. Here's what you need to know.on May 10, 2024 at 7:43 pm
How worried should parents be, and what symptoms should they watch out for? Doctors put the new data into perspective.
- First Opinion essay on colorectal cancer screening: letter and responseon May 10, 2024 at 5:00 pm
To encourage robust, good-faith discussion about difficult issues, STAT publishes selected Letters to the Editor received in response to First Opinion essays. Today’s edition is a bit different ...
- Colorectal Cancer Rates Falling in Older Adults but Rising in Childrenon May 9, 2024 at 8:49 pm
New research finds that while cases of colorectal cancer are declining in older adults, they have significantly increased in children.
via Bing News