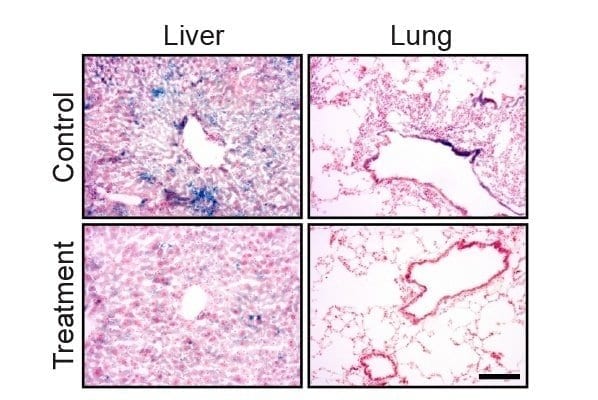
The results lay the foundation for researchers to plan a future phase 1 clinical trial to test the safety of the therapy in a small group of patients
Results from a recent preclinical study have shown that a new drug combination therapy being developed at Virginia Commonwealth University Massey Cancer Center effectively killed colon, liver, lung, kidney, breast and brain cancer cells while having little effect on noncancerous cells. The results lay the foundation for researchers to plan a future phase 1 clinical trial to test the safety of the therapy in a small group of patients.
“It is still too premature to estimate when a clinical trial will open to further test this drug combination therapy, but we are now in the planning phase and encouraged by the results of these laboratory experiments,” says Andrew Poklepovic, M.D., oncologist and member of the Developmental Therapeutics research program at VCU Massey Cancer Center and assistant professor in the Division of Hematology, Oncology and Palliative Care at VCU School of Medicine. “We are also encouraged by the fact that the drugs used in this therapy are either already approved by the FDA to treat certain cancers or are currently being investigated in other clinical trials.”
Featured in the journal Molecular Pharmacology, the study led by Paul Dent, Ph.D., demonstrated that the drugs sorafenib and regorafenib synergize with a class of drugs known as PI3K/AKT inhibitors to kill a variety of cancers. Sorafenib and regorafenib work by blocking the production of enzymes called kinases, which are vital to the growth and survival of cancer cells. Sorafenib is currently approved by the FDA to treat kidney and liver cancers, and regorafenib is currently approved for the treatment of colorectal cancer. However, sorafenib and regorafenib do not directly affect PI3K and AKT kinases, which are also very active in promoting cancer cell survival. The addition of a PI3K/AKT inhibitor to the combination of sorafenib and regorafenib dramatically increased cell death and was even effective against cells with certain mutations that make one or the other drug less effective.
“We know that there are certain cellular processes that are frequently dysregulated in cancers and important to cell proliferation and survival, but if you shut down one, then cells can often compensate by relying on another,” says Dent, Universal Corporation Distinguished Professor for Cancer Cell Signaling and member of the Developmental Therapeutics research program at VCU Massey Cancer Center as well as vice chair of the Department of Neurosurgery at VCU School of Medicine. “We are blocking several of these survival pathways, and the cancer cells are literally digesting themselves in an effort to stay alive.”
Go deeper with Bing News on:
Drug combination therapy
- FDA To Hold Advisory Committee Meeting On MDMA Therapy For PTSD, Welcomes Public Comment
Another significant move forward for MDMA-assisted therapy being made available to patients suffering from PTSD ...
- Bristol Myers Squibb Cancer-Treatment Trial Misses Endpoint
Bristol Myers Squibb said its trial evaluating a combination of cancer treatments failed to meet its primary endpoint.
- Keto Diet Boosts Lifesaving Antifungal Drug in Mice
It’s possible that a keto diet influences the gut microbiome in a way that changes how the drug is absorbed. The dietary shift could also boost the immune system, making it easier for the drug to ...
- Merck's endometrial cancer therapy fails trial
Merck said on Thursday its therapy did not meet the main goal of disease-free survival in certain patients with endometrial cancer during a late-stage trial.
- Ask the doctors: Medication, talk therapy may help with panic disorders
Many of us will experience a panic attack once or twice over the course of our lifetimes. But when they occur repeatedly, they fall into a category known as panic disorder. It is important to seek ...
Go deeper with Google Headlines on:
Drug combination therapy
[google_news title=”” keyword=”Drug combination therapy” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]
Go deeper with Bing News on:
Combination therapy
- FDA To Hold Advisory Committee Meeting On MDMA Therapy For PTSD, Welcomes Public Comment
Another significant move forward for MDMA-assisted therapy being made available to patients suffering from PTSD ...
- Combination immunotherapy treatment enhances immune response for people with malignant gliomas
Investigators at the UCLA Health Jonsson Comprehensive Cancer Center have pinpointed a combination immunotherapy treatment that enhances the immune response for people with malignant gliomas, an ...
- Merck's endometrial cancer therapy fails trial
Merck said on Thursday its therapy did not meet the main goal of disease-free survival in certain patients with endometrial cancer during a late-stage trial.
- Merck says Keytruda failed as first-line therapy for uterine cancer
Merck (NYSE:MRK) announced Thursday that its anti-PD-1 therapy Keytruda as a combination therapy failed to reach the main goal in a Phase 3 trial as a first-line treatment for patients with ...
- Phanes Therapeutics, Inc. Announces Clinical Supply Agreement with Roche to Evaluate PT217 in Combination with an anti-PD-L1 Therapy
Phanes Therapeutics, Inc. (Phanes), a clinical stage biotech company focused on innovative drug discovery and development in oncology, announced today that it has entered into a clinical supply ...
Go deeper with Google Headlines on:
Combination therapy
[google_news title=”” keyword=”combination therapy” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]